Anemia
W/U-cbc, T+C, PT/PTT, Lytes, UA, Retic, IFTs, B12/Folate, LDH, Haptoglobin, Plasma Hb, LFTs (Save a lav and red for hematology consult)
Decreased Production:
Low retic count (corrected retic=% retic x crit/45 % normal is 1-3%)
¯ MCH=hypochromia
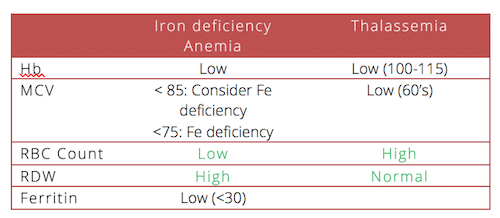
Use RDW to differentiate Iron defic from thal
Microcytic-defic or iron, globin (thal), or porphyrin (sideroblastic or lead poison)
Thal-¯ synthesis of globin chains, beta minor mcv<80
Sideroblastic-¯ porphyrin production. Increased serum iron. Can be idiopathic in the elderly try giving B6. Preleukemic state. Lead poisoning (basophilic stippling)
Normocytic-chronic disease or active loss
Macrocytic-
B12 Deficiency
Pernicious anemia
can be exacerbated by nitrous oxide (J Neurol Neurosurg Psychiatry 1998;64:563-564 Subacute combined degeneration of the spinal cord after nitrous oxide anaesthesia: role of magnetic resonance imaging)
takes 4 years to develop, paresthesias, propio or vibratory
Folate
Aplastic-myelophthisic (marrow replaced by tumor, etc.)
Increased Destruction
Intravascular-schizocyte
Extravascular-spherocyte
ferrous sulfate does not require vitamin c, only ferric
Oral Iron
Intravenous Iron
Iron sucrose
or
Benifor or Ferraheme
The Following is from EMCASES:
The WOMB Trial showed that young women can safely tolerate a hemoglobin as low as 50g/L
The WOMB trial was a multi-centered Dutch trial that enrolled 521 women with severe postpartum anemia (hemoglobin 48 to 79 g/L) who were randomized to transfusion or transfusion only if they developed severe symptoms. It found no differences in any important outcomes (recovery of hemoglobin, 6 week hemoglobin). There was, however, a non-clinically significant difference in fatigue scores at 7 days that was not persistent at later time point. 517 units were transfused to the “transfused group” vs. 88 for the group for only severe symptoms.
Allo-immunization is the most important and under-recognized risk associated with red cell transfusions
Perhaps the most important and under-recognized risk of red cell transfusions is allo-immunization among women of childbearing age.
Allo-imunization, which has a rate of 8% per transfusion in young women, involves the development of antibodies against red blood cells, which in future pregnancies can cross the placenta and precipitate hemolytic disease of the newborn in women who have received previous transfusion, can render the patient ineligible for an organ transplant if required and can make them unmatchable for future transfusions.
Think of a blood transfusion as a blood transplant: When you give someone a blood transfusion, you are changing their immune system for life. Red cell transfusions should not be thought of as a delivery system for iron!
Other risks of packed red blood cell transfusions include a 1/700 risk of TACO (Transfusion Associated Circulatory Overload) a 1/10,000 risk of TRALI (Transfusion Related Lung Injury) and a 1/40,000 risk of an acute hemolytic transfusion reaction.
Indications for Iron for anemia in Emergency Medicine
The American Society of Anesthesiologists recommend against RBC transfusions in young, healthy patients without ongoing blood loss and a hemoglobin >60 g/L, unless they are symptomatic or hemodynamically unstable. Symptoms to screen for include chest pain, SOB, pre-syncope, lightheadedness, hypotension and tachycardia.
Fatigue, pallor and reduced exercise tolerance are NOT, in and of themselves, an indication for red cell transfusion.
The trigger for transfusion related to “ongoing blood loss” will depend on acuity of blood loss, volume of ongoing bleeding and hemodynamic instability.
Indications for IV Iron:
- Oral iron poorly tolerated or failure of oral trial
- Poor oral absorption (ie. gastric bypass, celiac disease, gastritis)
- Rate of bleeding too brisk for oral iron
- Severe anemia (Hb <90g/L) especially if ongoing bleeding
- Time-sensitive pressures (OR etc.)
The main contraindications to IV Iron are active systemic infection (eg: suspected sepsis) since iron is a good microbial nutrient, and a known allergic or hypotensive reaction in the past.
Risks of IV Iron
- Hypotension (1-2%)
- Serious allergic reactions (< 1 in 1,000,000)
Other more common adverse reactions include joint aches, muscle cramps, headache, chest discomfort, nausea, vomiting and diarrhea, which generally resolve spontaneously within 24hrs of administration of IV iron.
Administering IV Iron for anemia in Emergency Medicine
The product you choose will depend on the dose you want to give, how quickly you want to deliver it and the side-effect profile (see order set example at end of summary)
| Iron Sucrose (Venofer) | Ferumoxytol (Feraheme) | |
| Dose (Max) | 300mg in 250mL NS | 510mg in 17mL (add to 50ml NS) |
| Infusion Time | 2 hrs | 15-60 mins |
| Serious Hypersensitivity | 0.6 per million | <5 in 1000 |
| Cost | $120 | $200 |
Patients with the following risk factors should receive slower infusions (e.g. Feraheme® [ferumoxytol] over 60 minutes or Venofer® [iron sucrose] 300mg over 2 hours)
- Age > 65 yrs
- Baseline systolic BP less than 100
- Severe asthma or eczema
- Severe respiratory or cardiac disease
- Treatment with beta-blockers, ACE inhibitors or 3 or more anti-hypertensive medications
- Nephrology patients
After IV Iron, and with ongoing oral supplementation, a patient’s hemoglobin will start to rise 3-7 days after the IV infusion. You can expect a 1-2 point rise in the hemoglobin per day, and after 2-4 weeks the hemoglobin will have risen 20-30g/L.
Sunnybrook Hospital IV Iron Indications & Criteria for use
Sunnybrook Hospital IV Iron order sheet
Oral Iron supplementation after IV Iron:
Ferrous sulfate 300mg 1 tab QHS: contains 60mg of elemental iron
- Take at bedtime on empty stomach at least 2 hours after meals with Vitamin C 500mg
- Avoid taking with calcium or magnesium supplements as these decrease absorption.
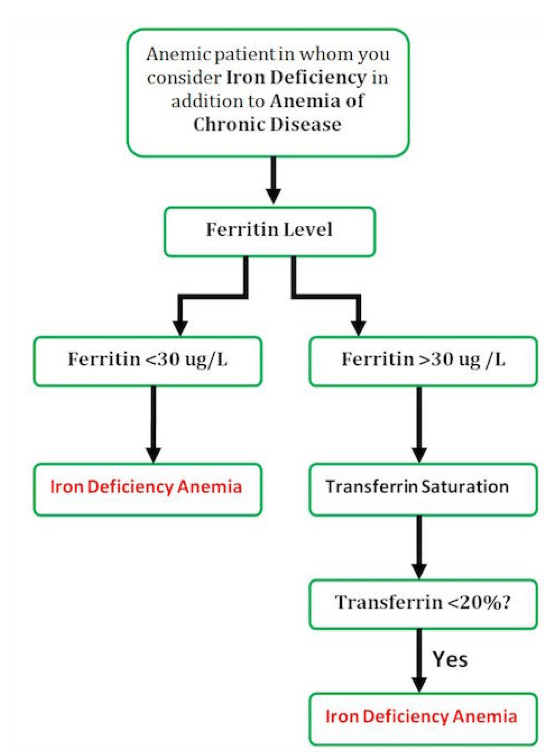
Lab Interpretation to determine iron deficiency
Patients with thalassemia may also have iron deficiency contributing to their anemia. If a patient with thalassemia has a Ferritin <30, consider them iron deficient.
IV Iron instead of recurrent red cell transfusions for anemia of chronic disease
Would you transfuse this patient with RBCs?
Most of the patients in the ED found to be anemic are elderly. These patients generally fall into one of three categories: One third will have a simple nutritional deficiency (iron or B12). One third will have anemia secondary to a chronic disease. The rest of the patients will have an undifferentiated cause of their anemia that will require further investigation.
For an elderly patient with multiple comorbidities it can be challenging to determine if their anemia is secondary to iron deficiency as well as anemia of chronic disease. To you help differentiate and decide whether a patient would benefit from IV iron and supplementation, our experts have suggested the following approach:
Which pre-operative patients require red cell transfusions?
IV Iron for anemia in pre-operative patients?
The FOCUS trial sought to determine whether a higher threshold for blood transfusion would improve recovery in patients who had undergone surgery for a hip fracture. They showed that even among elderly patients with known coronary artery disease or multiple coronary risk factors, there was less mortality post-operatively at 30 and 90 days among patients with a transfusion trigger of 80 g/L compared to those with a higher transfusion trigger.
Observational studies of the use IV iron pre-operatively for patients with anemia have shown a reduced rate of red cell transfusion required.
Patient hand-out for iron therapy from Sunnybrook Hospital (with permission)
Key References
Weiskopf, R et al.,Human Cardiovascular and Metabolic Response to Acute, Severe Isovolemic Anemia.JAMA. 1998;279(3):217-221.
Litton E, Xiao J, Ho KM. Safety and efficacy of intravenous iron therapy in reducing requirement for allogeneic blood transfusion: systematic review and meta-analysis of randomised clinical trials. BMJ. 2013;347:f4822.
Prick BW, Steegers EA, Jansen AJ, et al. Well being of obstetric patients on minimal blood transfusions (WOMB trial). BMC Pregnancy Childbirth. 2010;10:83.
Carson JL, Terrin ML, Noveck H, et al. Liberal or restrictive transfusion in high-risk patients after hip surgery. N Engl J Med. 2011;365(26):2453-62.
Munoz M et al. Very-short-term perioperative intravenous iron administration and postoperative outcome in major orthopedic surgery: a pooled analysis of observational data from 2547 patients. Transfusion. Dec 27, 2012.
Himmel’s Top 20 Do’s and Don’ts for Anemia in the ED (from EMCases)
1. Don’t give needless blood. Young women who are healthy can tolerate Hb of down to 50g/L if bleeding has stopped or menorrhagia is managed. Older folks (>80g/L) with multiple risk factors for heart disease do tolerate Hb of 80g/L.
2. Anemia is NOT good but Don’t give lots of blood as this does not help the underlying problem – blood is a Band-Aid solution.
3. Don’t forget that some one has to look at the etiology at some point.
4. Don’t miss thalassemia. It is wide spread.
5. Do give good advice regarding oral iron. Once per day, on an empty stomach with water (tons of patients are still told to take it with food to minimize GI side effects, believe me (I ask all the time).
6. Do give vitamin C 500mg along with oral Iron to maximize absorption in the gut.
7. Don’t forget that iron deficiency causes lots of problems other than anemia such as cognitive impairment and, in children, learning problems.
8. Do look at the RDW and the RBC count to determine whether a patient has iron deficiency anemia or thalassemia or something else.
9. Do communicate to the Family MD, or consultant, or clinic.
10. You would be amazed as to how many patients are NOT prescribed FE for Iron deficiency. Do discuss compliance and do not assume someone else will do it.
11. Do arrange follow-up. Some patients may require repeated doses of IV iron.
12. Don’t forget that iron may cause a positive stool for occult blood.
13. Do order a serum ferritin when you are not sure about the diagnosis of iron deficiency anemia. Serum ferrtin < 30 is a sensitive and specific marker for iron deficiency anemia BUT not in people with inflammation and especially NOT in people with chronic renal disease. These patients may be iron deficient and have ferritin of >100.
14. Don’t give blood just because the patient gets blood every month. Seniors from nursing homes with iron deficiency anemia need a systems approach. After the 2nd or 3rd visit to the ED for blood some one has to come up with a plan.
15. In those with inflammation (high hepcidin level), oral iron may not work and they may still benefit from IV iron. This is probably NOT a decision for the ED but someone has to arrange the referral. Don’t leave it up to someone else.
16. Pearl: in the elderly with anemia (perhaps 15 % of all elderly): 1/3 deficiency (fe, b12, folate),1/3 inflammation, and 1/3 unknown. Some one has to arrange a follow-up. Don’t ignore the anemia.
17. With iron deficiency and a HG < 90g/L (this is very significant anemia for the long term although it generally does not require transfusion), the patient needs to about 1000mg or more of absorbed ELEMENTAL iron plus daily requirement. This will take 6 months or more. Iron sulfate is only 30 % elemental iron and only 10% is absorbed if not less in ill patients. So, it takes time and persistence. Do counsel patients that it may take months of treatment.
18. Each group needs to think this out. Giving iron blindly is not good; giving blood blindly is really bad. Do meet with your ED group and hematologists and come up with an a agreed upon protocol.
19. If the patient is really anemic (<90g/L) and iron deficient, I have NO problem giving Iron sucrose in the ED (300 mg) and arranging a follow-up. This is the right thing to do. If you choose to send them to a clinic, Do warn them of the cost. Clearly, this is new knowledge for the community and will really require educational and political influence and power.
20. Remember, you may get some hostile push-back from those who sent the patients for BLOOD and from the patients who may believe that they will die without two or three units of blood. Do take time for good patient-centered care.
Anemia and Iron Deficiency in Critical Illness
Hemolysis
Direct antiglobulin test (DAT) and indirect antiglobulin test (IAT). AHG = antihuman globulin. A. The DAT reflects in vivo antibody sensitization of erythrocytes. Erythrocytes are washed to remove any unbound antibodies, and anti-IgG AHG reagent is then added. IgG antibodies cannot cause direct erythrocyte agglutination, but if the erythrocytes are coated with IgG antibodies, the AHG reagent will cause them to agglutinate. This test can also be performed using anti-complement AHG reagent. If IgG antibodies are present, they can be eluted off the erythrocytes for specificity determination. B. The IAT is used to detect the presence of IgG antibodies in serum (in vitro sensitization). Reagent erythrocytes are incubated in the presence of serum that potentially contains antibodies. If antibodies are present, they bind to their target antigens on the reagent erythrocytes. After the incubation period the erythrocytes are washed to remove unbound antibodies. Anti-IgG AHG reagent is added and will cause IgG-coated erythrocytes to agglutinate.
The indirect antiglobulin test and its clinical applications
The IAT is used by the blood bank to detect unexpected erythrocyte antibodies in the patient’s serum or plasma. The IAT is thefinal phase of the antibody screen and serologic crossmatchprocedures. In an antibody screen the recipient’s serum is incubatedwith 2 or 3 different type O erythrocytes that express clinicallysignificant antigens. This erythrocyte and serum mixture isthen incubated with anti-IgG AHG and observed for agglutination.If agglutination occurs, the screen is considered positive,and further testing is performed to determine the specificityof the IgG antibody. The serologic crossmatch procedure is alsoan IAT procedure and is performed on patients with unexpectederythrocyte antibodies to verify that the potential donor erythrocyteunit lacks the antigen corresponding to the recipient’s antibody.In this procedure, the patient’s serum is incubated with erythrocytesfrom a potential erythrocyte donor unit, and anti-IgG reagentis added. Agglutination would indicate an incompatibility betweenthe donor erythrocytes and the recipient’s IgG antibody. Thusthe results of the IAT reflect in vitro erythrocyte sensitization.The anti-complement AHG reagent is generally not used in antibodyscreening or crossmatch procedures.
Sickle Cell Anemia
Polycythemia
Crit>60%, start to worry about viscosity induced problems
Itching after baths
WBC Disorders
African Americans have decreased white counts
Chapter 121-Disorders of Hemostasis
Thrombin Sensitive-I,V,VIII,XIII
K Sensitive-II, VII, IX, X
Heparin Sensitive-Iia, Ixa, Xa, Platelet Factor 3
Platelet Disorders-ptechiae and mucosal bleeding
Coag Disorders-joint or muscle bleeding
Bleeding Time-tests just platelet function
PT-extrinsic and common, PTT-intrinsic and common
Neutrophilia
Because glucocorticoids induce prompt eosinopenia and basophilopenia, these cells are almost universally absent in the blood of the acutely injured or infected patient. Thus, their presence should indicate that (1) the acutely ill patient may have concomitant adrenocortical insufficiency, (2) the neutrophilia derives from the inappropriate production of GM-CSF (e.g., by malignant cells), or (3) the neutrophilia is one manifestation of a hematopoietic neoplasm (a chronic myeloproliferative disorder, myelodysplastic syndrome, lymphoma, or acute nonlymphocytic leukemias associated with eosinophilia).
Acute Intermittent Porphyria
(AIP)
Get single void urine and have lab check for porphobilinogen, preferably using Trace PBG Kit. Must eliminate comperting substances like urobilinogen before using Ehrlich’s reagent. (Ann Intern Med 2005;142(6):439)
AIP is an autosomal dominant disorder that results from an approximately 50% deficiency of Porphobilinogen deaminase (PBG).
The majority of subjects with PBG deaminase deficiency remain asymptomatic.
Most drugs that are harmful in AIP induce hepatic ALA synthase and cytochrome P-450 enzymes. Sulfonamide antibiotics are not inducers and may inhibit PBG deaminase. Reduced caloric and carbohydrate intake enhances the induction of ALA synthase in animals and in AIP can increase ALA and PBG and precipitate symptoms. Administration of carbohydrate can reduce hepatic ALA synthase and P-450 enzymes.
The mechanism of neural damage in AIP is unknown. Porphyrias and related disorders associated with increased ALA have similar neurologic manifestations. ALA is structurally analogous to gamma-aminobutyric acid (GABA) and can interact with GABA receptors. However, ALA and other products of the heme pathway have not been convincingly shown to be neurotoxic. The suggestion that heme deficiency may occur in nervous tissue in these disorders is also unproved.
Symptoms rarely occur before puberty and seldom if ever recur throughout adult life. Characteristically, attacks last for several days or longer, often require hospitalization, and are followed by complete recovery.
Abdominal pain is the most common symptom, is usually steady and poorly localized, but may be cramping.
Tachycardia, hypertension, restlessness, fine tremors, and excess sweating may be due to sympathetic overactivity.
Other manifestations include nausea and vomiting; constipation; pain in the limbs, head, neck, or chest; muscle weakness; and sensory loss. Ileus with distention and decreased bowel sounds is common. However, increased bowel sounds and diarrhea may be seen. Because the abdominal symptoms are neurologic rather than inflammatory, tenderness, fever, and leukocytosis are generally absent or mild. Dysuria and bladder dysfunction may occur. Recurrent attacks tend to be similar in a given patient.
Peripheral neuropathy in AIP is primarily motor, results from axonal degeneration, and does not develop in all patients with acute attacks, even when abdominal symptoms are severe. Rarely, neuropathy develops apart from abdominal symptoms. Weakness most commonly begins in proximal muscles (often requiring a careful examination to detect) and more often in the arms than the legs. It can be asymmetric and focal. Tendon reflexes may be little affected or hyperactive in the early stages but are usually decreased or absent with advanced neuropathy. Cranial and sensory nerves can be affected. Progression to respiratory and bulbar paralysis and death seldom occurs unless the porphyria is not recognized, the use of harmful drugs is not discontinued, and appropriate treatment is not instituted. Sudden death, presumably from cardiac arrhythmia, may also occur.
The central nervous system can be involved. Anxiety, insomnia, depression, disorientation, hallucinations, and paranoia, which can be especially severe during acute attacks, may suggest a primary mental disorder or hysteria.
Seizures may occur as an acute neurologic manifestation of AIP, as a result of hyponatremia, or secondary
to causes unrelated to porphyria. Hyponatremia may be due to hypothalamic involvement and inappropriate antidiuretic hormone secretion; vomiting, diarrhea, and poor intake; or excess renal sodium loss.
After several days, an attack may resolve quite rapidly, with abdominal pain disappearing within a few hours and paresis within a few days. Attacks during the luteal phase of the menstrual cycle usually resolve with the onset of menses. Even advanced neuropathy is potentially reversible. Pain, depression, and other symptoms are sometimes chronic.
Chronic hepatic abnormalities are common in AIP, and affected patients have an increased risk of hepatocellular carcinoma (apparently not associated with hepatitis B or C). AIP may predispose to chronic hypertension and be associated with impaired renal function. The mechanisms of these associations are unknown.
Barbiturates and sulfonamides are the most notorious drugs to induce an attack
Reduced caloric intake is also a common cause
Heme therapy and carbohydrate loading are specific therapies because they repress hepatic ALA synthase and overproduction of ALA and PBG. Heme therapy is most effective in this regard and should be initiated early, but only after the diagnosis of a porphyric attack is confirmed by a marked increase in urinary PBG. Diagnosis is more difficult after heme therapy, which can at least transiently normalize ALA and PBG.
The standard regimen for heme therapy is 3-4 mg heme per kilogram body weight infused intravenously once daily for 4 days. A longer course of treatment is seldom necessary if treatment is started early. Efficacy is reduced and recovery less rapid when treatment is delayed and neuronal damage is more advanced. It is not effective for chronic symptoms of AIP. A lyophilized hematin (hydroxyheme) preparation is available in the United States. The manufacturer recommends reconstitution with sterile water. However, the product is unstable and degradation products adhere to endothelial cells, platelets, and coagulation factors and cause a transient anticoagulant effect and phlebitis at the site of infusion. Reconstitution with human albumin enhances the stability of hematin and prevents these side effects. Heme arginate, which is available in Europe and South Africa, is much more stable than hematin and also does not have these side effects. It is an investigational drug in the United States.
Carbohydrate loading may suffice for mild attacks and can be given orally as sucrose, glucose polymers, or carbohydrate-rich foods. If oral intake is poorly tolerated or is contraindicated by distention and ileus, intravenous administration of glucose (at least 300 g daily) is usually indicated. A central venous line facilitates more complete parenteral nutrition support and avoids excess fluid volumes. Parenteral nutrition support may be indicated in some patients who require heme therapy.
Treatment of seizures is problematic because almost all antiseizure drugs can exacerbate AIP. Bromides, gabapentin, and probably vigabatrin can be given safely. beta-Adrenergic blocking agents may control tachycardia and hypertension in acute attacks of porphyria, but they may be hazardous in patients with hypovolemia, in whom increased catecholamine secretion may be an important compensatory mechanism. Numerous other therapies have been tried in this disease but have not been consistently useful.