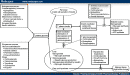
Euglycemia in the ICU
Non-critical patient goals
110 pre-meal, 180 post-meal
Lantus 0.2 units/kg
Insulin aspart 4 units with each meal
151-180 1
181-200 2
201-250 3
251-300 5
301-350 6
351-400 8
- Krinsley J. Decreased mortality of critically ill patients with the use of an intensive glycemic management protocol. Crit Care Med. 2003;31:A19.
- Van den Berghe GH, Wouters P, Weekers F, et al. Intensive insulin therapy in the critically ill patients. N Engl J Med. 2001;345:1359-1367. Abstract
- Krinsley JS. Association between hyperglycemia and increased hospital mortality in a heterogeneous population of critically ill patients. Mayo Clin Proc. 2003;78:1471-1478. Abstract
- Coursin DB, Murray MJ. How sweet is euglycemia in critically ill patients. Mayo Clin Proc. 2003;78:1460-1462. Abstract
- Finney SJ, Zekveld C, Elia A, Evans TW. Glucose control and mortality in critically ill patients. JAMA. 2003;290:2041-2047. Abstract
Krinsley JS. Effect of an intensive glucose management protocol on the mortality of critically ill adult patients. Mayo Clin Proc 2004 Aug; 79:992-1000
I would propose the following guidelines for a SSI protocol. Comments welcome. 1. There will be a three-tiered sliding scale dosage algorithm (see below). Unless otherwise indicated, an order to simply “start SSI protocol” would mandate that the nurses start with low-dose algorithm. If desired by the physician, a higher dose algorithm could be selected via an order to “start medium-dose SSI protocol”, or “start high-dose SSI protocol”, or “start SSI protocol using high-dose regimen”, or something to that effect. 4. If the fourth or any subsequent POC is > 200, the nurse will automatically move up to the next highest SSI dosage algorithm. At that point, the count will restart, and if the fourth or any subsequent POC thereafter is > 200, the nurse will automatically move up again to the next highest SSI dosage algorithm. 5. If, after starting the high-dose regimen, the fourth or any subsequent POC is > 200, the physician will be notified. A custom regimen can then be ordered, if so desired. 6. A one-third or greater cut in enteral or parenteral nutrition rate will cause the nurse to reduce the SSI regimen to the low-dose protocol, and restart the count from there (in regards to items 4. and 5., above). 7. The three SSI dosage regimens will be as follows:
POC
Low dose
Medium-dose
High-dose
130-160 –>
2
4
6
161-200 –>
4
6
8
201-240 –>
6
8
10
241-280 –>
8
10
12
> 280 –>
10
12
14
8. For hypoglycemia: POC 80-90 –> recheck POC in three hours POC 70-79 –> Give half amp D50 and recheck POC in 2 hrs POC < 70 –> Give one amp D50 and recheck POC in 2 hrs
Review of why it works (Curr Opin Crit Care 2005;11:304)
Article saying it is sugar control, not insulin (Crit Care Med 2003;31:359)
SR of lit on how to get it done (Crit Care 2006;10:R19)
continuous infusion works best
New Van den Berghe Article
Large prospective intention to treat trial by Van den Berghe (NEJM 2006;354(5):449) showed no mortality benefit at all; when subgroup of patients in ICU > 72 hrs examined, then mortality benefit and morbidity benefit returned
The electronic appendix to the article is very worrisome. The figures for hypoglycemia (<2,2 mmol/L) are not what we are used to find in the literature and not in line with her first study. There were 3,1% hypoglycemia in the conventional arm of the intention to treat group and 18,7% in the intensive group and there were 3,9% events in the conventional arm of the <3 days group and 25,1% in the intensive group… Remember that the VISEP trial was stopped after recruiting 488 out of 600 patients “because of no difference in mortality (21.9 vs. 21.6%, p = 1.0) and frequent hypoglycemia in the intensive insulin therapy arm (12.1 vs. 2.1%, ” Dr Van denBerghe states that nurses used a modified titration guideline for this study, I think that part of the problem lies in the protocol used at least for hypoglycemias… In the meantime I think I will stick with my strict glucose control protocol and wait for the NICE-SUGAR trial to answer my questions
Cost
Cost effectiveness of intensive glycemic control (Chest 2006;129:644)
Conclusions In this large, international, randomized trial, we found that intensive glucose control increased mortality among adults in the ICU: a blood glucose target of 180 mg or less per deciliter resulted in lower mortality than did a target of 81 to 108 mg per deciliter. (ClinicalTrials.gov number, NCT00220987 [ClinicalTrials.gov] .) (NICE NEJM Volume 360:1283-1297March 26, 2009
Does intensive insulin therapy really reduce mortality in critically ill surgical patients? a reanalysis of meta-analytic data
Jan O Friedrich , Clarence Chant and Neill KJ Adhikari
Critical Care 2010, 14:324doi:10.1186/cc9240
Published: 21 October 2010
Abstract (provisional)
Two recent systematic reviews evaluating intensive insulin therapy (IIT) in critically-ill patients grouped randomized controlled trials (RCTs) by type of intensive care unit (ICU). The more recent review found that IIT reduced mortality in patients admitted to a surgical ICU, but not in those admitted to medical or mixed medical-surgical ICUs, or in all patients combined. Our objective was to determine whether IIT saves lives in critically-ill surgical patients regardless of type of ICU. Pooling mortality data from surgical and medical subgroups in mixed-ICU RCTs (16 trials) with RCTs conducted exclusively in surgical ICUs (5 trials) and medical ICUs (5 trials), respectively, showed no effect of IIT in the subgroup of surgical patients (RR 0.85 [95% CI 0.69-1.04, P=0.11]; I2=51% [95% CI 1-75%]) or medical patients (RR 1.02 [95% CI 0.95-1.09, P=0.61]; I2=0% [95% CI 0-41%]). There was no differential effect between subgroups (interaction P=0.10). There was statistical heterogeneity in the surgical subgroup, with some trials demonstrating significant benefit and others demonstrating significant harm, but no surgical subgroup consistently benefited from IIT. Such a re-analysis suggests that IIT does not reduce mortality in critically ill surgical or medical patients. Further insights may come from individual patient data meta-analyses or from future large multicentre RCTs in more narrowly defined subgroups of surgical patients.