Recommendations of American Academy of CCM (Crit Care Med 2008;36:953)
Medical Futility
If you had a cold and went to an HD center offering $1000 to get dialyzed, would we do it? Atkinson et al: Identification of Futility in Intensive Care” Lancet, Vol. 344, pp. 1203-1206, 1994. Crippen D, Burrows R: Medically inappropriate medical care of moribund patients: a critical care physicians perspective. Critical Care Forum 1997: 1 (2)f1-f5. Tilden VP et al: Decisions about life-sustaining treatment. Impact of physicians’ behaviors on the family. Arch Intern Med 1995 Mar 27;155(6):633-638 Ayres, SM: Who Decides When Care is Futile?. Hospital Practice, Vol. 26, pp. 41-53, 1991. Murphy DJ. Barbour E. GUIDe (Guidelines for the Use of Intensive Care in Denver): a community effort to define futile and inappropriate care. New Horizons. 2(3):326-31, 1994 Aug. Brain dead=dead Pneumonia=the old man’s friend Criteria for Medical Futility (Youngner 1988) 1. It will not work 2. Works in a physiologic sense, but does not prolong time till death (2-3 days is not prolonging) the above our Our choice 3. No quality of life 4. Unlikely but possible chance of success Last two are Their choice Coin operated ventilator dying is a process with a certain outcome, but a unknown point of irreversibility Prolong Life, not dying Three Options 1. DNR 2. No escalation 3. Treatment withdrawal Consider setting a maximum physiologic dose of pressors admission with reservations (admit to crit care just to determine neuro reversibility) MRI, EEG, Somatosensory evoked potentials administer narcotics before neuro exam if opioids were given dnr tells us what we can not do, not what we must do. Now, take the poor patient dying of metastatic colon cancer, languishing on a floor with a 1:6 or higher nurse-patient ratio. He needs pain control, his diarrhea tended to, etc., etc., etc. – he may even need a final good bath. Now, I can consult, make recommendations, and try to follow up on the medical ward while tending to 15 – 20 critically patients 5 floors away in the ICU, or bring him to the ICU for intensive medical, nursing and ethical management. Claudia, I am not necessarily making lemonade, because a “penny of prevention is worth a pound of cure.” We treat this person for 24 hours in the ICU, get appropriate pain, medical, nursing and ethical issues addressed – and this guarantees that he will never be admitted after a cardiac arrest, intubated, in shock to languish for days until the family gets over the anger, hostility and rage than these issues were not addressed before hand. BTW, the nursing staff does not object because that realize the wisdom of this prophylactic approach. (Mark Mazer) Thank you for codifying this. In Hospice we called it the “Daughter from California” syndrome. The child with the most emotional baggage moved the furthest away, has had the least to do with the client and thus has the highest guilt level. Usually the youngest child, often ‘daddy’s baby girl.’ Crippen’s Law of the Obstinant Outlier “In any family consensus, there will always be one outlier that will want to do something different. That person will always live furthest from the patient and had not seen them for the longest time. They usually live in California.”
Death Notification
(Acad Emerg Med 2005;12(4):296) Ggather; gather the family; ensure that all members are present. Rresources; call for support resources available to assist the family with their grief, i.e., chaplain services, ministers, family, and friends. Iidentify; identify yourself, identify the deceased or injured patient by name, and identify the state of knowledge of the family relative to the events of the day. Eeducate; briefly educate the family as to the events that have occurred in the emergency department, educate them about the current state of their loved one. Vverify; verify that their family member has died. Be clear! Use the words “dead” or “died.” _space; give the family personal space and time for an emotional moment; allow the family time to absorb the information. Iinquire; ask if there are any questions, and answer them all. Nnuts and bolts; inquire about organ donation, funeral services, and personal belongings. Offer the family the opportunity to view the body. Ggive; give them your card and access information. Offer to answer any questions that may arise later. Always return their call. Hobgood et al.’s GRIEV_ING Protocol for Death Notification G (G)ather the familyIdentify next of kin, surrogates R (R)esourcesAssemble critical personnel (e.g., social worker, chaplain) I (I)ntroduceProvide introductions for staff, family members E (E)ducateSummarize prehospital and ED events V (V)erifyState that the patient died _ (Pause)Allow for emotional reaction I (I)nviteInvite, gather, and share information N (N)uts and boltsReview important next steps G (G)iveGive condolences, contact information Seven Legal myths about end-of-life care (JAMA 2000;284(19):2495) Illness trajectory (BMJ 2005;330:1007) 


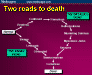 Crippen’s Article on why we screwed up end of life decisions (Critical Care 2005;9:317-8) Some reasons why this occurs are as follows: 1. Physicians tell surrogates that they can make any decision they want as an open-ended ideal. This puts them in the position of being buyers in a consumer’s market. By asking them to make a choice, they imply that their authority to make choices extends to making bad ones. 2. Moribund patients look comfortable on ‘life support’. An observer’s primal reaction to the vibrant external appearance of a body supported in an intensive care unit (ICU) is radically different from that to a corpse on a morgue slab [9]. As long as the patient ‘looks viable, it is emotionally easier to accept the pie in the sky bye and bye long shot cure’. If the patient can just be maintained comfortably for long enough, then a cure may eventually become possible. 3. Surrogates dislike being in a position
Crippen’s Article on why we screwed up end of life decisions (Critical Care 2005;9:317-8) Some reasons why this occurs are as follows: 1. Physicians tell surrogates that they can make any decision they want as an open-ended ideal. This puts them in the position of being buyers in a consumer’s market. By asking them to make a choice, they imply that their authority to make choices extends to making bad ones. 2. Moribund patients look comfortable on ‘life support’. An observer’s primal reaction to the vibrant external appearance of a body supported in an intensive care unit (ICU) is radically different from that to a corpse on a morgue slab [9]. As long as the patient ‘looks viable, it is emotionally easier to accept the pie in the sky bye and bye long shot cure’. If the patient can just be maintained comfortably for long enough, then a cure may eventually become possible. 3. Surrogates dislike being in a position
of making decisions that directly result in the death of a loved one. Once life-supporting care is instituted, the patient has options for ‘survival’ that they did not have before, even though they are dependent on ‘life support’. There are now variables that decision makers control, and it is much easier to avoid decisions that may hasten death [10]. Instead of yielding to inevitable death, the potential now exists to manipulate it. Life support generates an outcome that is no longer inevitably fatal. 4. Physicians do not have an exceptional track record in explaining end-of-life issues to patients and their families [11]. It is not uncommon for physicians to ask loaded questions in their quest for end-of-life decisions. For example, ‘This is your grandmother’s 17th transfer from a skilled nursing facility in 3 months for sepsis and respiratory failure, and now she’s in kidney failure as well. What do you want to do: everything or let her die?’ Given that choice, most surrogates would opt for doing something rather than nothing, even if ‘something’ perpetuated open-ended pain and discomfort. 5. The popular media, especially the tabloids, frequently feature anecdotal articles describing patients who have awakened after years of coma [12]. Most if not all of these patients’ conditions have been embellished to generate public interest, and frequently subsequent investigators cannot find these patients. Accordingly, some families feel that if life support systems can maintain vital signs for a day or a week, then ‘suspended animation’ should be possible indefinitely, until a cure is found. 6. The notion of ‘medical futility’ as an end-stage process in which vital signs cannot be supported further is poorly understood by both physicians and surrogates [13]. In fact, any medical treatment capable of sustaining hemodynamics, ventilation, and metabolism is not technically futile if it achieves that limited goal [14]. A treatment is futile only if it is unsuccessful in achieving a stated goal. Therefore, if a patient in a progressive, inevitable death spiral is placed on mechanical ventilation, it is not technically futile if vital signs are sustained, however briefly. It is medically inappropriate but not technically futile. Under the current rules, the only test of futility is that embodied by the question, ‘Will this treatment result in sustained life?’ If the answer is ‘yes’, then virtually any treatment is fair game, even if it will do nothing to revitalize the patient. colleagues have a name for those relatives that always seem to come from California: seagulls. They fly in, crap on everything, and fly away. Tips on communicating at end-of-life Trial of intensive therapies medical futility (adjective); the process whereby, through technological means, diverse tissues and organs are maintained in a prolonged state of dying and decompositiion in a gruesome simulation of what was once a human life. Examples: See Ariel Sharon, Randal McCloy, Terri Schiavo. –M Darwin Medicine in the U.S. is in worsening trouble. Tort reform is only a small piece. IMHO, getting Americans to understand that: (1) Death is NOT optional (2) They can only have the extraordinary health care they can afford this is the place a HUGE amount of money is spent for no legitimate reason. (3) There isnt enough money in the world to let every American have any health care they think they should have. (4) Insurance is a contract to pay for certain things, NOT to pay for all imaginable health care. Personally, I think all insurance for people with average or better incomes should REQUIRE a 20% co-pay to decrease unnecessary care. (5) Health care providers have the courage, and the legal basis, to tell patients No, that isnt appropriate and I will not order it. will be necessary before things change.
managing death in the icu
j randall curtis from paternalism to autonomy the right to die autonomy by surrogate from purple dots to dnr (La Guardia Hospital in Queens) slow codes honest prognostication clarifying confusing distinctions Heroic <> ordinary Withhold<>Withdraw Killing<>Let die Overruling Autonomy Allocation of Scarce Resources Futility Scopolamine Patch 1.5 mg Review of Withdrawal of Treatment in the ED (Acad Emerg Med 2006;13:1328) Current Anaesthesia & Critical Care Volume 17, Issue 5 , 2006, Pages 255-262 Futility and the critically ill adult patient: A framework  J.F. Cosgrovea, , , I.D. Nesbitta and C. Bartleyb In critical care medicine the prevailing approach is to stabilize and support patients whilst recovery occurs i.e. diagnostic and curative. However with intensive care mortality rates at 1822%1 care of the dying is inevitable. Add to this an in-hospital mortality of 3040% and it becomes obvious that palliative care models are necessary if intensive care doctors are to function effectively. Keenan demonstrated that 70% of intensive care deaths are associated with the withdrawal or withholding of life-sustaining treatments.2 If the original intention was to save and cure changing to palliation from active treatment may be counter-intuitive for doctors working in acute hospital medicine with access to modern technology.3 and 4 Personal communication with senior intensive care clinicians throughout the UK has cited examples of consultations with the seriously ill and their relatives where the possibility of death had never previously been raised by healthcare staff. Thus in addition to actually providing effective palliative care, the critical care doctor must be skilled in preparing patients and their families for the possibility of a loved one dying. This article provides guidance for moving from a curative to a palliative care model within the medico-legal and ethical standards of UK society. If decisions about futility are to be made doctors must consider what margins of error are acceptable. Some may believe that they always make correct decisions about discontinuing treatment but perfection is unlikely as the only means of ensuring that active treatment is never stopped inappropriately is to continue treating patients with no chance of survival. This approach would undoubtedly produce conflict as it could cause harm and suffering to a dying patient and impact on other patients and allocation of resources. Thus whilst clinicians strive to achieve 100% sensitivity and 100% specificity the reality is that no doctor is capable of such judgement. However there is a duty to achieve consensus. With respect to end of life care, assessment and management centres on distinguishing who will survive and who will not. Each clinician has a point at which they are sure a patient will not survive, with some more decisive than others. A degree of uncertainty is inevitable but consistency in decision-making across institutions and indeed the profession can minimize conflict. Recognizing futility Resources in an advanced medical service tend to be deployed in the pursuit of the preservation of life.4 However if the costs of this policy become insupportable there is a need to identify dying patients and care for them accordingly.5 In the UK the General Medical Council advises that we must recognize progression of a patient’s condition when death is approaching6 and whilst the term futility may be associated with conflicts between paternalistic staff and demanding relatives it is an unavoidable concept. A diagnostic framework is therefore helpful for its use in a palliative care context that allows doctors to be clear about the legalities and ethics of such decision making. In its simplest form (i.e. a dictionary definition) futility is something that is useless or ineffective. In the medical context futile treatment is care that does not accomplish its intended purpose.7 This intended purpose has been further refined by Jecker and Pearlman producing four definitions7: 1. Treatment that does not offer a reasonable chance of survival. 2. Treatment that was either useless or ineffective. 3. Treatment that fails to offer a minimum quality of life or modicum of medical benefit. 4. Treatment that cannot possibly achieve patients goals. Definitions (1) and (2) are definitions of quantitative or physiological futility, and relate to the physical effects of treatment. They apply to alterations in organ function, and are arguably the least complicated and least controversial for physicians to understand and utilize. Definitions (3) and (4) are qualitative and more holistic, looking for a benefit to the patient. Others, beyond the clinical team have a stake in determining normative futility. Treatment that does not offer a reasonable chance of survival Individual clinicians inevitably regard reasonable differently, nevertheless (regardless of such opinions) the process involves marrying basic clinical impressions to information linking predicted mortality with the pathophysiological severity of illness e.g. physiological scoring systems such as APACHE (Acute Physiology and Chronic Health evaluation) II and III and SAPS (Sequential Acute Physiology Score), the numbers of body organs in failure, types of body organs in failure etc.8, 9 and 10 This is useful and often very accurate but must be taken in the context of being based upon populations and clearly should not be applied in isolation to individuals. Conversely (hence differences in the meaning of reasonable) patient assessment can also include known historical reports of successful treatment in similar patients. Again however we are applying data from external sources to individuals and must be very certain that it is robust, applicable and current e.g. there is data demonstrating that patients with liver failure plus sepsis plus ventilatory failure, who then develop renal failure, die without a liver transplant. It could therefore be used to decide that renal replacement therapy is futile in patients for whom a liver transplant is not possible.11 However, since this data was published, the medical management of hepatic, and especially hepato-renal failure, has changed. Populations of patients with alcohol-induced liver failure have also changed12 (younger patients, more female patients etc.) Can historical data relating to middle-aged male patients be applied to a 22-year old female if no such patients were included in the dataset? And if anecdotal data exists citing survival of such a patient is it totally applicable to a new critically ill individual?. The same is true of case reports about critically ill patients because they may not be the same as the critically ill patient currently occupying the hospital bed. A debate is necessary (see below). Treatment that is either useless or ineffective This has the benefit of hindsight, although at face value for individual patients it will appear unhelpful. However, if there is doubt or disagreement over prognosis and the likely effectiveness of a proposed treatment, then a time-limited trial of the new therapy may be useful. Clear (physiological) target parameters with specific review times can be agreed e.g. PaO2, blood pressure etc. and/or independence from specified forms of organ support in n hours/days time. This clarity is essential to enable agreement at the end of the trial of therapy over its success or failure. Setting so-called SMART targets can be helpful: they should be Specific, Measurable (rather than a nebulous improvement), Agreed (caring team, family, patient), Realistic (to potentially demonstrate this measured improvement) and within an appropriate Timescale. Examples include: Reductions in ventilator pressures. Improved alveolar-arterial oxygen gradients. White cell count to fall below 20×109/l or normothermia after 72 h of new anti-microbial therapy. This approach can confirm to all involved that continued treatment is indeed futile, and reassure concerned parties that everything has been done. It entails continued use of expensive resources and requires a co-ordinated multidisciplinary approach. Treatments failing to offer a modicum of medical benefit, or achieving patients goals Normative futility definitions necessitate decisions about quality of life and best interests as viewed by the patient. Ideally this should be discussed with a competent patient. Unfortunately this is frequently impossible in critical care. More complex debates involving staff and family are therefore necessary and include addressing the value of continued treatment to the patient, deciding whether on-going management should be active or palliative. Thus, deciding upon futility may be progressive rather than sudden as information is collected to determine best interests and quality of life. Such quality of life decisions must be legal, ethical and acceptable in today’s society. Legal and ethical considerations overlap. Legal considerations Patients should be treated in their best interests. With an unconscious emergency patient in the absence of futility or a pre-stated proxy decision an assumption of best interests is that the patient would wish to survive and active treatment would be continued/commenced. If the patient is conscious and orientated their consent to treatment must be given with management options being presented to the patient. They can consent to or refuse various treatments accordingly. Medical staff must presume capacity until proven otherwise and it should be strongly emphasized that patients are not to be treated as lacking capacity if they make a decision that doctors believe to be unwise e.g. refusal of life-saving surgery. Treatment under such circumstances constitutes assault. In adult critical care however many critically ill patients fall between full mental capacity and complete unconsciousness. The Mental Capacity Act of 2005 is the most recent attempt to clarify this area.13 and 14 Patients are deemed to lack capacity if they have a disturbance or impairment of the mind or brain that prevents them from making independent decisions. Such incapacity can be either temporary e.g. sepsis, drug-induced, or permanent e.g. dementia. To assess capacity, the doctor must ascertain whether the patient can understand and retain relevant information, weigh pros and cons and communicate the decision. They must consider all relevant circumstances and take the following steps: Does potential for capacity in relation to the matter in question exist? If potential for future capacity exists, when is it likely to be? As far as is reasonably practicable, doctors must permit, encourage or improve patients ability to participate fully in acts or decisions. This can range from active verbal encouragement, to formal counselling, revision of sedative requirements and dialysis to remove substances contributing to sedation e.g. urea. Where determining such capacity relates to life-sustaining treatments doctors must not, in considering whether treatment are in a patient’s best interests, be motivated by a desire to bring about his/her death. As far as is reasonably ascertainable doctors must consider: ○ Patients past and present wishes and feelings, in particular, any relevant written statement made during a period of capacity. ○ Beliefs and values likely to influence decisions if capacity existed. ○ Any other factors that would be considered if the patient had capacity. The next part of the Act is perhaps the most radical, since for the first time in the UK, it allows for one adult to make treatment decisions on behalf of another adult. Doctors determining best interests must take into account the views of: Anyone named by the patient as someone to be consulted on the matter in question. Anyone engaged in caring for the patient or interested in their welfare. Anyone with lasting power of attorney granted by the person. Any deputy appointed for the person by the court. The Act builds in some safeguards e.g. if in doubt treatment is continued until a decision from the courts is available. Strict conditions also apply to those with lasting power of attorney. Most importantly they cannot authorize refusal of, or consent to the institution or continuation of life-sustaining treatment unless such matters were expressly stated when the Power of Attorney was given by the individual who is now the patient. Deputies nominated by the court can also make decisions on a patient’s behalf. The scope of these decisions is specific and determined by the court e.g. they could give or withhold consent for insertion of a percutaneous tracheostomy to facilitate ventilatory weaning, but they could not refuse ventilation to sustain life. Additional considerations may relate to any Advance Decisions15 that can refuse treatment if: At the time of making an advance decision the person making it was over 18 years of age and had full capacity. It applies to circumstances that arise with specified proposed treatments e.g. a wish not to undergo cardiopulmonary resuscitation in the event of an unwitnessed, in-hospital cardiac arrest. The person now lacks capacity. There are no reasonable grounds to believe that new circumstances/developing treatments now exist (unanticipated by the patient) that would now alter their decision to refuse treatment. Stricter criteria apply to the critical care situations where an advance decision does not apply to life-sustaining treatment unless: The patient specified that the decision was to apply even if life is at risk! The advance decision is in writing, and signed by the patient or by another person in the patient’s presence and at his/her direction, and any signature is witnessed. Whilst an appropriate advance decision to refuse treatment should be viewed as equivalent to a contemporaneous, informed refusal of treatment, doctors will not incur liability for continuing to treat until they are sure that such a valid, applicable advance decision exists. If necessary this can involve recourse to the courts. Other legal considerations include the European Convention for Human Rights16 (English Law since 1998) with three articles immediately relevant to critical care: Article 2: with the exception of the prevention of criminal acts or lawful use of a death penalty, everyone’s right to life shall be protected. Article 3: no one shall be subjected to torture or to inhuman or degrading treatment or punishment. Article 8: with respect to an individual within the norms of a democratic society an organization cannot interfere with their rights unless it is to uphold the law or protect the rights and freedoms of others. These are essentially core tenets of medical ethics and are considered further below. Bioethical considerations Formulating a framework for treatment decisions can be aided by an awareness of the four bioethical principles described by Beauchamp and Childress17: beneficence, non-maleficance, distributive justice and autonomy i.e. doing good, avoidance of harm, accounting for the greater good and ascertaining and acting in accordance with the patient’s wishes. The potential for conflict is inevitable necessitating discussion. Beneficence vs. non-maleficance Does multi-organ support benefit the patient or will its burden cause unnecessary pain and suffering, and be degrading or disrespectful? Although individual life is protected by law (Article 2), is life-sustaining treatment appropriate if a patient is dying? I.e. other best interests such as the goals of a good death have potentially become more important than preserving life in the physiological context. In the face of an overwhelming certainty of death there is clearly a mandate to ensure a patient is respected, free from distress and receives appropriate levels of analgesia in accordance with Article 3 of the Human Rights Act. Respect for best interests is also inter-twined with Article 8. These applications of Articles 3 and 8 are the basis of effective palliative care. Such therapy may hasten a patient’s death, but ethically this predictable but non-intentional effect (the Doctrine of Double Effect or DDE) is permissible if the primary effect is beneficial and more important than the secondary effect. The DDE is discussed in greater depth elsewhere but its key elements are: The intention is to produce a good effect e.g. effective analgesia. The good effect is not achieved through the bad effect i.e. death follows effective analgesia as opposed to death precipitating effective analgesia. There is sufficient reason to permit the bad effect i.e. the benefit of the good (effective palliative care) outweighs the bad (death). Autonomy When attempting to diagnose futility and institute palliative care staff must consider both of the following: Does initiating palliative care interfere with a patient’s rights and freedoms? Does continuing active treatments interfere with rights and freedoms? A responsibility therefore exists to determine patients autonomous wishes. Much of this process has been discussed previously but other factors to consider include: Potential for causing harm (physical/psychological) through such discussions. Clinicians duty to the critically ill patient vs. duty to avoid/minimize harm to the loved ones of patients. At present it is not expected that families will take responsibility for end-of-life decisions as it may not be ethical to expect relatives to live with such decisions. They do however contribute to the process and at times are capable of providing clinicians with accurate information in relation to a patient’s autonomous wishes18 but as a counter-point clinicians must also be aware that relatives may be unable to separate their own wishes from the patient’s likely autonomous wishes e.g. to see an end to perceived suffering, to prolong life-continuing treatments or (rarely) to have an ulterior motive associated with the patient’s death. Occasions also arise when doctors considering a diagnosis of futility are faced with what they regard as extraordinary requests from patients or relatives i.e. a reasonable treatment must have a reasonable efficacy and thus not be futile. Currently negative demands e.g. refusal of active interventions outweigh positive ones e.g. a request for organ transplant in futile situations. This principle aids decision-making, resource allocation and distributive justice (see below). Deciding what constitutes reasonable treatment can be controversial and situations may arise where clinicians have to resort to the courts to decide upon appropriate levels of care e.g. Portsmouth NHS Trust vs. the parents of Charlotte Wyatt.19 Distributive justice This is where continuing with active but futile treatment of a dying patient influences the treatment or potential treatment of others, e.g. a deteriorating patient with several days of multi-organ dysfunction occupies a bed on a full critical care unit. In the same hospital patients are scheduled for life-saving surgery. Their operations must either be postponed due to lack of a critical care bed or they must be transferred from the operating theatre to another hospital’s intensive care unit. Is it ethical to expose one patient to the potential harm of an inter-hospital transfer because there is a bed-shortage exacerbated by continuing futile treatment of another patient? Once again debate is necessary with guidance being sought from society as a whole. Achieving consensus Even with adherence to this ethico-legal framework conflict can occur. Clear communication and documentation is essential if controversy is to be minimised.20 Each case requires clinicians to maintain an open mind regardless of their initial reading of a situation.21 Agreed, written guidelines for treatment withdrawal based on GMC and BMA guidance have the potential to smooth this area of conflict.6, 20, 21 and 22 They can be applied and subject to audit. It is also sensible to ensure that at least two consultants are involved in the decision making process and are in agreement with withdrawal decisions. The multidisciplinary team caring for the patient must agree and all decisions must be documented in the case notes. Levels of care forms are useful for communicating and clarifying such decisions.22, 23 and 24 A patient can thus be deemed to be for all active treatments, active treatments except those specified e.g. renal replacement therapy, escalation of inotropes above predetermined levels etc. for active treatment but not CPR or for end of life care only. Such forms (Fig. 1) should include the following details: Who has made the decision (two named clinicians)? What was the basis of the decision e.g. the patient’s wishes, futility, etc. Whether the patient and his family are aware of the decision, and if not, why not? The expectation being that patient or family should always be involved. Date for review of the decision. (219K) Figure 1. Withdrawal of active care form. Such a system is more sensitive and specific than the usual Do Not Resuscitate (DNR) orders. It can also add to clarity of communication for staff covering out of hours emphasizing the painstakingly agreed approach to a patient’s care in the event of deterioration or failure to respond to treatment. They can also prevent the unfortunate assumption that DNR implies that a patient is not for active treatment at any point during their hospital stay. If consensus cannot be reached, a time-limited trial of treatment (discussed above) might be of use. Seeking a second opinion from an appropriate clinician may help when disagreement occurs and in the USA, hospital ethics committees are involved in complex withdrawal decisions.25 Studies of the impact of these committees have not shown prolongations in intensive care length of stay where treatment discontinuation was negotiated compared to other methods of withdrawal decisions.25 In the UK, Clinical Ethics Committees have the potential for an increasing role in this area.26 and 27 Finally if resolution is impossible further advice and opinion should be sought from other independent authorities including the High Court e.g. Miss B in 2002, Wyatt vs. Portsmouth NHS Trust in 2004, Mr. A in 2005 and Baby MB in March 2006.18, 28, 29, 30 and 31 Summary and conclusions In conclusion palliative models of care are necessary for good practice in the critical care environment. It is essential that clinicians appreciate the concept of futility in both its physiological and normative forms. In doing so they should also possess background knowledge of the legal and bioethical issues surrounding end of life care and have the skills to communicate these issues to others i.e. patients, relatives and staff. By doing so, they will improve the provision of effective end of life care. References 1 D. Bennett and J. Bion, ABC of intensive care: organisation of intensive care, BMJ 318 (1999) (7196), pp. 14681470. Abstract + References in Scopus | Cited By in Scopus 2 S.P. Keenan, K.D. Busche, L.M. Chen, L. McCarthy, K.J. Inman and W.J. Sibbald, A retrospective review of a large cohort of patients undergoing the process of withholding or withdrawal of life support, Crit Care Med 25 (1997) (8), pp. 13241331. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 3 M.A. DeVita, J. Groeger and R. Truog, Current controversies in critical care ethics: not just end of life, Crit Care Med 31 (2003) (5 Suppl), p. S343. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 4 D. Callahan, Living and dying with medical technology, Crit Care Med 31 (2003) (5 Suppl), pp. S344S346. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 5 Slater E. Severely malformed children: wanteda new approach. BMJ 1973; 285. 6 General Medical Council, Withholding and withdrawing life-prolonging treatments: good-practice in decision making, GMC, London (2002). 7 N.S. Jecker and R.A. Pearlman, Medical futility. Who decides?, Arch Intern Med 152 (1992) (6), pp. 11401144. Abstract + References in Scopus | Cited By in Scopus 8 C.M. Watts and W.A. Knaus, The case for using objective scoring systems to predict intensive care unit outcome, Crit Care Clin 10 (1994) (1), pp. 7389 [discussion 912]. 9 R.B. Becker and J.E. Zimmerman, ICU scoring systems allow prediction of patient outcomes and comparison of ICU performance, Crit Care Clin 12 (1996) (3), pp. 503514. Abstract | Abstract + References in Scopus | Cited By in Scopus 10 D.H. Beck, B.L. Taylor, B. Millar and G.B. Smith, Prediction of outcome from intensive care: a prospective cohort study comparing acute physiology and chronic health evaluation II and III prognostic systems in a United Kingdom intensive care unit, Crit Care Med 25 (1997) (1), pp. 915. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 11 P.S. Kamath, R.H. Wiesner, M. Malinchoc, W. Kremers, T.M. Therneau and C.L. Kosberg et al., A model to predict survival in patients with end-stage liver disease, Hepatology 33 (2001) (2), pp. 464470. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 12 L. Donaldson, On the state of public health: the annual report of the Chief Medical Officer, Department of Health, London (2001). 13 Mental Capacity Act 2005. HMSO. 14 S.M. White and T.J. Baldwin, The Mental Capacity Act 2005implications for anaesthesia and critical care, Anaesthesia 61 (2006) (4), pp. 381389. Full Text via CrossRef 15 Age Concern, October 2005. Advance statements, advance directives and living wills. 16 The European Convention on Human Rights 1998; Articles 2, 3 and 8. 17 T. Beauchamp and J. Childress, Principles of biomedical ethics (4th ed), Oxford University Press, Oxford (1994). 18 J. Rogers, S. Ridley, P. Chrispin, H. Scotton and D. Lloyd, Reliability of the next of kins estimates of critically ill patients quality of life, Anaesthesia 52 (1997) (12), pp. 11371143. Abstract + References in Scopus | Cited By in Scopus 19 Portsmouth NHS Trust vs. Wyatt & Ors, EWHC 2247, 2004 (Fam.). 20 British Medical Association, Withholding and withdrawing life-prolonging medical treatment, BMJ Books, London (1999). 21 J. Way, A.L. Back and J.R. Curtis, Withdrawing life support and resolution of conflict with families, BMJ 325 (2002) (7376), pp. 13421345. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 22 P. Levack, Live and let die? A structured approach to decision-making about resuscitation, Br J Anaesth 89 (2002) (5), pp. 683686. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 23 R.D. Troung and J.P. Burns, Excellence in end-of-life care: a goal for intensivists, Intensive Care Med 28 (2002) (9), pp. 11971199. 24 I.D. Nesbitt and D.W. Ryan, End of life care in ICU, Care of the Critically Ill 21 (2005), pp. 99100. Abstract + References in Scopus | Cited By in Scopus 25 M.D. Dowdy, C. Robertson and J.A. Bander, A study of proactive ethics consultation for critically and terminally ill patients with extended lengths of stay, Crit Care Med 26 (1998) (2), pp. 252259 [Erratum in: Crit Care Med 1998 Nov;26(11):1923]. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 26 The Ethox Centre. www.ethox.org.uk. 27 A.M. Slowther and T. Hope, Clinical ethics committees, BMJ 321 (2000) (7262), pp. 649650. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 28 B v An NHS Trust, EWHC 429, 2002 (Fam.). 29 C. Dyer, Woman makes legal history in right to die case, BMJ 324 (2002) (7338), p. 629. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 30 An NHS Trust v A & Anor (adult patient: withdrawal of medical treatment) EWCA 1145, 2005 (Civ.). 31 An NHS Trust v MB & Anor, EWHC 507, 2006 (Fam.). Non-invasive for end of life (Crit Care Med 2008;36:789) From: Stephen Streat (ADHB) <StephenS@adhb.govt.nz> Date: Sun, May 10, 2009 at 10:56 PMSubject: Re: [CCM-L] persistent myoclonus To: Discussion of Critical Care Medicine <ccm-l@ccm-l.org>I’ve been off-line for a few days (conference in Sydney), but would like to respond to this because I think that there is one very neat treatment for this condition — which we use frequently, think is superior to everything else on several grounds and I can recommend — thiopentone infusion.Before you dismiss this post — let me explain …If this truly is post-anoxic status myoclonus, in a situation where there has been a profound severe hypoxic-ischaemic insult (e.g. prolonged arrest, delayed CPR etc etc), then as Michaal and others have pointed out, it is a VERY bad sign and forms a strong part of a decision to withdraw treatment. Of course it needs to be distinguished from transient intermittent seizures and from the Lance-Adams syndrome, which do not share the dire prognostic significance of status myoclonus in this context. But I’m assuming that someone has done that and that you are dealing with “the real thing”.First thing to say is “treating it with curative intent” is a strategy which is doomed to fail. Don’t try. All you will do is produce either a delayed death, or another Schiavo (see attached PDF, read the first few pages especially).Second thing — if status myoclonus is “bad” (as it often is) it is awful to look at — for both the healthcare workers and the patients family. Sometimes (I’ve seen it), it can cause heatstroke ! (in the patient). I’ve seen one patient recently (in whom I let it go on for 30 minutes to show a “blind” family what I was talking about) who developed a fever (42C), profound shock and a lactate of 20 mmol/l in just 30 minutes. I actually reversed all this (noradrenaline, 3 litres of iced-saline, thiopentone, external cooling) just to keep the patient alive for the next few hours, so that I could hold another couple of family meetings, including waiting for another family member to arrive by plane.Third thing — WHEN you have decided that “treatment with curative intent” is not appropriate, you then have the issue of “how do I palliate this awful phenomenon”. This is what thiopentone infusion does — it controls the phenomenon very well, without much of a “loading dose”, and with usually only a modest (150-250 mg/hr) infusion rate. It also does this with remarkable haemodynamic stability (you have probably already volume loaded the patient well, prior to the decision to provide symptomatic control only). It does not stop the patient from breathing (after all, the brain stem is usually fairly well preserved). So — you can (and we do) then wean the patient from IPPV to spontaneous breathing, and then extubate them and pull out all the lines (ETT, NG. art line, cease all monitoring) keeping only one line for thiopentone, and a Foley catheter ..The patient looks peaceful (and is), is devoid of NG and ETT (so their face looks “near-normal”) and there is no ventilator, ECG, art-line or pulse-oximetry — only the family, the nurse, conversation, weeping, singing, sometimes some music, a few drinks (yes reader, a few drinks), laughter and comfort — and no evident distress, from the patient, family or nurses/doctors. Meanwhile — thiopentone is cumulating all the time that myoclonus is being controlled. Typically the patients die a few hours after extubation.Typically I might chart “Thiopentone, 150mg/hr by infusion + 100mg IV prn for control of status myoclonus” and review the infusion rate every few hours. If the patient is getting a lot of prn, I might write “Thiopentone 300mg/hr plus 50mg IV prn.” Nurses are very happy with this. Cheap, effective monotherapy with good symptom control, allows for highly “low-tech” good comfort care and peaceful death. Very smooth. No panic moments. Comes recommended with 4 or 5 pale yellow stars. No RCTs. YMMD.Comments ?Perhaps I should write up a small case series of ten or so to raise peoples awareness of this option.
J.F. Cosgrovea, , , I.D. Nesbitta and C. Bartleyb In critical care medicine the prevailing approach is to stabilize and support patients whilst recovery occurs i.e. diagnostic and curative. However with intensive care mortality rates at 1822%1 care of the dying is inevitable. Add to this an in-hospital mortality of 3040% and it becomes obvious that palliative care models are necessary if intensive care doctors are to function effectively. Keenan demonstrated that 70% of intensive care deaths are associated with the withdrawal or withholding of life-sustaining treatments.2 If the original intention was to save and cure changing to palliation from active treatment may be counter-intuitive for doctors working in acute hospital medicine with access to modern technology.3 and 4 Personal communication with senior intensive care clinicians throughout the UK has cited examples of consultations with the seriously ill and their relatives where the possibility of death had never previously been raised by healthcare staff. Thus in addition to actually providing effective palliative care, the critical care doctor must be skilled in preparing patients and their families for the possibility of a loved one dying. This article provides guidance for moving from a curative to a palliative care model within the medico-legal and ethical standards of UK society. If decisions about futility are to be made doctors must consider what margins of error are acceptable. Some may believe that they always make correct decisions about discontinuing treatment but perfection is unlikely as the only means of ensuring that active treatment is never stopped inappropriately is to continue treating patients with no chance of survival. This approach would undoubtedly produce conflict as it could cause harm and suffering to a dying patient and impact on other patients and allocation of resources. Thus whilst clinicians strive to achieve 100% sensitivity and 100% specificity the reality is that no doctor is capable of such judgement. However there is a duty to achieve consensus. With respect to end of life care, assessment and management centres on distinguishing who will survive and who will not. Each clinician has a point at which they are sure a patient will not survive, with some more decisive than others. A degree of uncertainty is inevitable but consistency in decision-making across institutions and indeed the profession can minimize conflict. Recognizing futility Resources in an advanced medical service tend to be deployed in the pursuit of the preservation of life.4 However if the costs of this policy become insupportable there is a need to identify dying patients and care for them accordingly.5 In the UK the General Medical Council advises that we must recognize progression of a patient’s condition when death is approaching6 and whilst the term futility may be associated with conflicts between paternalistic staff and demanding relatives it is an unavoidable concept. A diagnostic framework is therefore helpful for its use in a palliative care context that allows doctors to be clear about the legalities and ethics of such decision making. In its simplest form (i.e. a dictionary definition) futility is something that is useless or ineffective. In the medical context futile treatment is care that does not accomplish its intended purpose.7 This intended purpose has been further refined by Jecker and Pearlman producing four definitions7: 1. Treatment that does not offer a reasonable chance of survival. 2. Treatment that was either useless or ineffective. 3. Treatment that fails to offer a minimum quality of life or modicum of medical benefit. 4. Treatment that cannot possibly achieve patients goals. Definitions (1) and (2) are definitions of quantitative or physiological futility, and relate to the physical effects of treatment. They apply to alterations in organ function, and are arguably the least complicated and least controversial for physicians to understand and utilize. Definitions (3) and (4) are qualitative and more holistic, looking for a benefit to the patient. Others, beyond the clinical team have a stake in determining normative futility. Treatment that does not offer a reasonable chance of survival Individual clinicians inevitably regard reasonable differently, nevertheless (regardless of such opinions) the process involves marrying basic clinical impressions to information linking predicted mortality with the pathophysiological severity of illness e.g. physiological scoring systems such as APACHE (Acute Physiology and Chronic Health evaluation) II and III and SAPS (Sequential Acute Physiology Score), the numbers of body organs in failure, types of body organs in failure etc.8, 9 and 10 This is useful and often very accurate but must be taken in the context of being based upon populations and clearly should not be applied in isolation to individuals. Conversely (hence differences in the meaning of reasonable) patient assessment can also include known historical reports of successful treatment in similar patients. Again however we are applying data from external sources to individuals and must be very certain that it is robust, applicable and current e.g. there is data demonstrating that patients with liver failure plus sepsis plus ventilatory failure, who then develop renal failure, die without a liver transplant. It could therefore be used to decide that renal replacement therapy is futile in patients for whom a liver transplant is not possible.11 However, since this data was published, the medical management of hepatic, and especially hepato-renal failure, has changed. Populations of patients with alcohol-induced liver failure have also changed12 (younger patients, more female patients etc.) Can historical data relating to middle-aged male patients be applied to a 22-year old female if no such patients were included in the dataset? And if anecdotal data exists citing survival of such a patient is it totally applicable to a new critically ill individual?. The same is true of case reports about critically ill patients because they may not be the same as the critically ill patient currently occupying the hospital bed. A debate is necessary (see below). Treatment that is either useless or ineffective This has the benefit of hindsight, although at face value for individual patients it will appear unhelpful. However, if there is doubt or disagreement over prognosis and the likely effectiveness of a proposed treatment, then a time-limited trial of the new therapy may be useful. Clear (physiological) target parameters with specific review times can be agreed e.g. PaO2, blood pressure etc. and/or independence from specified forms of organ support in n hours/days time. This clarity is essential to enable agreement at the end of the trial of therapy over its success or failure. Setting so-called SMART targets can be helpful: they should be Specific, Measurable (rather than a nebulous improvement), Agreed (caring team, family, patient), Realistic (to potentially demonstrate this measured improvement) and within an appropriate Timescale. Examples include: Reductions in ventilator pressures. Improved alveolar-arterial oxygen gradients. White cell count to fall below 20×109/l or normothermia after 72 h of new anti-microbial therapy. This approach can confirm to all involved that continued treatment is indeed futile, and reassure concerned parties that everything has been done. It entails continued use of expensive resources and requires a co-ordinated multidisciplinary approach. Treatments failing to offer a modicum of medical benefit, or achieving patients goals Normative futility definitions necessitate decisions about quality of life and best interests as viewed by the patient. Ideally this should be discussed with a competent patient. Unfortunately this is frequently impossible in critical care. More complex debates involving staff and family are therefore necessary and include addressing the value of continued treatment to the patient, deciding whether on-going management should be active or palliative. Thus, deciding upon futility may be progressive rather than sudden as information is collected to determine best interests and quality of life. Such quality of life decisions must be legal, ethical and acceptable in today’s society. Legal and ethical considerations overlap. Legal considerations Patients should be treated in their best interests. With an unconscious emergency patient in the absence of futility or a pre-stated proxy decision an assumption of best interests is that the patient would wish to survive and active treatment would be continued/commenced. If the patient is conscious and orientated their consent to treatment must be given with management options being presented to the patient. They can consent to or refuse various treatments accordingly. Medical staff must presume capacity until proven otherwise and it should be strongly emphasized that patients are not to be treated as lacking capacity if they make a decision that doctors believe to be unwise e.g. refusal of life-saving surgery. Treatment under such circumstances constitutes assault. In adult critical care however many critically ill patients fall between full mental capacity and complete unconsciousness. The Mental Capacity Act of 2005 is the most recent attempt to clarify this area.13 and 14 Patients are deemed to lack capacity if they have a disturbance or impairment of the mind or brain that prevents them from making independent decisions. Such incapacity can be either temporary e.g. sepsis, drug-induced, or permanent e.g. dementia. To assess capacity, the doctor must ascertain whether the patient can understand and retain relevant information, weigh pros and cons and communicate the decision. They must consider all relevant circumstances and take the following steps: Does potential for capacity in relation to the matter in question exist? If potential for future capacity exists, when is it likely to be? As far as is reasonably practicable, doctors must permit, encourage or improve patients ability to participate fully in acts or decisions. This can range from active verbal encouragement, to formal counselling, revision of sedative requirements and dialysis to remove substances contributing to sedation e.g. urea. Where determining such capacity relates to life-sustaining treatments doctors must not, in considering whether treatment are in a patient’s best interests, be motivated by a desire to bring about his/her death. As far as is reasonably ascertainable doctors must consider: ○ Patients past and present wishes and feelings, in particular, any relevant written statement made during a period of capacity. ○ Beliefs and values likely to influence decisions if capacity existed. ○ Any other factors that would be considered if the patient had capacity. The next part of the Act is perhaps the most radical, since for the first time in the UK, it allows for one adult to make treatment decisions on behalf of another adult. Doctors determining best interests must take into account the views of: Anyone named by the patient as someone to be consulted on the matter in question. Anyone engaged in caring for the patient or interested in their welfare. Anyone with lasting power of attorney granted by the person. Any deputy appointed for the person by the court. The Act builds in some safeguards e.g. if in doubt treatment is continued until a decision from the courts is available. Strict conditions also apply to those with lasting power of attorney. Most importantly they cannot authorize refusal of, or consent to the institution or continuation of life-sustaining treatment unless such matters were expressly stated when the Power of Attorney was given by the individual who is now the patient. Deputies nominated by the court can also make decisions on a patient’s behalf. The scope of these decisions is specific and determined by the court e.g. they could give or withhold consent for insertion of a percutaneous tracheostomy to facilitate ventilatory weaning, but they could not refuse ventilation to sustain life. Additional considerations may relate to any Advance Decisions15 that can refuse treatment if: At the time of making an advance decision the person making it was over 18 years of age and had full capacity. It applies to circumstances that arise with specified proposed treatments e.g. a wish not to undergo cardiopulmonary resuscitation in the event of an unwitnessed, in-hospital cardiac arrest. The person now lacks capacity. There are no reasonable grounds to believe that new circumstances/developing treatments now exist (unanticipated by the patient) that would now alter their decision to refuse treatment. Stricter criteria apply to the critical care situations where an advance decision does not apply to life-sustaining treatment unless: The patient specified that the decision was to apply even if life is at risk! The advance decision is in writing, and signed by the patient or by another person in the patient’s presence and at his/her direction, and any signature is witnessed. Whilst an appropriate advance decision to refuse treatment should be viewed as equivalent to a contemporaneous, informed refusal of treatment, doctors will not incur liability for continuing to treat until they are sure that such a valid, applicable advance decision exists. If necessary this can involve recourse to the courts. Other legal considerations include the European Convention for Human Rights16 (English Law since 1998) with three articles immediately relevant to critical care: Article 2: with the exception of the prevention of criminal acts or lawful use of a death penalty, everyone’s right to life shall be protected. Article 3: no one shall be subjected to torture or to inhuman or degrading treatment or punishment. Article 8: with respect to an individual within the norms of a democratic society an organization cannot interfere with their rights unless it is to uphold the law or protect the rights and freedoms of others. These are essentially core tenets of medical ethics and are considered further below. Bioethical considerations Formulating a framework for treatment decisions can be aided by an awareness of the four bioethical principles described by Beauchamp and Childress17: beneficence, non-maleficance, distributive justice and autonomy i.e. doing good, avoidance of harm, accounting for the greater good and ascertaining and acting in accordance with the patient’s wishes. The potential for conflict is inevitable necessitating discussion. Beneficence vs. non-maleficance Does multi-organ support benefit the patient or will its burden cause unnecessary pain and suffering, and be degrading or disrespectful? Although individual life is protected by law (Article 2), is life-sustaining treatment appropriate if a patient is dying? I.e. other best interests such as the goals of a good death have potentially become more important than preserving life in the physiological context. In the face of an overwhelming certainty of death there is clearly a mandate to ensure a patient is respected, free from distress and receives appropriate levels of analgesia in accordance with Article 3 of the Human Rights Act. Respect for best interests is also inter-twined with Article 8. These applications of Articles 3 and 8 are the basis of effective palliative care. Such therapy may hasten a patient’s death, but ethically this predictable but non-intentional effect (the Doctrine of Double Effect or DDE) is permissible if the primary effect is beneficial and more important than the secondary effect. The DDE is discussed in greater depth elsewhere but its key elements are: The intention is to produce a good effect e.g. effective analgesia. The good effect is not achieved through the bad effect i.e. death follows effective analgesia as opposed to death precipitating effective analgesia. There is sufficient reason to permit the bad effect i.e. the benefit of the good (effective palliative care) outweighs the bad (death). Autonomy When attempting to diagnose futility and institute palliative care staff must consider both of the following: Does initiating palliative care interfere with a patient’s rights and freedoms? Does continuing active treatments interfere with rights and freedoms? A responsibility therefore exists to determine patients autonomous wishes. Much of this process has been discussed previously but other factors to consider include: Potential for causing harm (physical/psychological) through such discussions. Clinicians duty to the critically ill patient vs. duty to avoid/minimize harm to the loved ones of patients. At present it is not expected that families will take responsibility for end-of-life decisions as it may not be ethical to expect relatives to live with such decisions. They do however contribute to the process and at times are capable of providing clinicians with accurate information in relation to a patient’s autonomous wishes18 but as a counter-point clinicians must also be aware that relatives may be unable to separate their own wishes from the patient’s likely autonomous wishes e.g. to see an end to perceived suffering, to prolong life-continuing treatments or (rarely) to have an ulterior motive associated with the patient’s death. Occasions also arise when doctors considering a diagnosis of futility are faced with what they regard as extraordinary requests from patients or relatives i.e. a reasonable treatment must have a reasonable efficacy and thus not be futile. Currently negative demands e.g. refusal of active interventions outweigh positive ones e.g. a request for organ transplant in futile situations. This principle aids decision-making, resource allocation and distributive justice (see below). Deciding what constitutes reasonable treatment can be controversial and situations may arise where clinicians have to resort to the courts to decide upon appropriate levels of care e.g. Portsmouth NHS Trust vs. the parents of Charlotte Wyatt.19 Distributive justice This is where continuing with active but futile treatment of a dying patient influences the treatment or potential treatment of others, e.g. a deteriorating patient with several days of multi-organ dysfunction occupies a bed on a full critical care unit. In the same hospital patients are scheduled for life-saving surgery. Their operations must either be postponed due to lack of a critical care bed or they must be transferred from the operating theatre to another hospital’s intensive care unit. Is it ethical to expose one patient to the potential harm of an inter-hospital transfer because there is a bed-shortage exacerbated by continuing futile treatment of another patient? Once again debate is necessary with guidance being sought from society as a whole. Achieving consensus Even with adherence to this ethico-legal framework conflict can occur. Clear communication and documentation is essential if controversy is to be minimised.20 Each case requires clinicians to maintain an open mind regardless of their initial reading of a situation.21 Agreed, written guidelines for treatment withdrawal based on GMC and BMA guidance have the potential to smooth this area of conflict.6, 20, 21 and 22 They can be applied and subject to audit. It is also sensible to ensure that at least two consultants are involved in the decision making process and are in agreement with withdrawal decisions. The multidisciplinary team caring for the patient must agree and all decisions must be documented in the case notes. Levels of care forms are useful for communicating and clarifying such decisions.22, 23 and 24 A patient can thus be deemed to be for all active treatments, active treatments except those specified e.g. renal replacement therapy, escalation of inotropes above predetermined levels etc. for active treatment but not CPR or for end of life care only. Such forms (Fig. 1) should include the following details: Who has made the decision (two named clinicians)? What was the basis of the decision e.g. the patient’s wishes, futility, etc. Whether the patient and his family are aware of the decision, and if not, why not? The expectation being that patient or family should always be involved. Date for review of the decision. (219K) Figure 1. Withdrawal of active care form. Such a system is more sensitive and specific than the usual Do Not Resuscitate (DNR) orders. It can also add to clarity of communication for staff covering out of hours emphasizing the painstakingly agreed approach to a patient’s care in the event of deterioration or failure to respond to treatment. They can also prevent the unfortunate assumption that DNR implies that a patient is not for active treatment at any point during their hospital stay. If consensus cannot be reached, a time-limited trial of treatment (discussed above) might be of use. Seeking a second opinion from an appropriate clinician may help when disagreement occurs and in the USA, hospital ethics committees are involved in complex withdrawal decisions.25 Studies of the impact of these committees have not shown prolongations in intensive care length of stay where treatment discontinuation was negotiated compared to other methods of withdrawal decisions.25 In the UK, Clinical Ethics Committees have the potential for an increasing role in this area.26 and 27 Finally if resolution is impossible further advice and opinion should be sought from other independent authorities including the High Court e.g. Miss B in 2002, Wyatt vs. Portsmouth NHS Trust in 2004, Mr. A in 2005 and Baby MB in March 2006.18, 28, 29, 30 and 31 Summary and conclusions In conclusion palliative models of care are necessary for good practice in the critical care environment. It is essential that clinicians appreciate the concept of futility in both its physiological and normative forms. In doing so they should also possess background knowledge of the legal and bioethical issues surrounding end of life care and have the skills to communicate these issues to others i.e. patients, relatives and staff. By doing so, they will improve the provision of effective end of life care. References 1 D. Bennett and J. Bion, ABC of intensive care: organisation of intensive care, BMJ 318 (1999) (7196), pp. 14681470. Abstract + References in Scopus | Cited By in Scopus 2 S.P. Keenan, K.D. Busche, L.M. Chen, L. McCarthy, K.J. Inman and W.J. Sibbald, A retrospective review of a large cohort of patients undergoing the process of withholding or withdrawal of life support, Crit Care Med 25 (1997) (8), pp. 13241331. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 3 M.A. DeVita, J. Groeger and R. Truog, Current controversies in critical care ethics: not just end of life, Crit Care Med 31 (2003) (5 Suppl), p. S343. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 4 D. Callahan, Living and dying with medical technology, Crit Care Med 31 (2003) (5 Suppl), pp. S344S346. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 5 Slater E. Severely malformed children: wanteda new approach. BMJ 1973; 285. 6 General Medical Council, Withholding and withdrawing life-prolonging treatments: good-practice in decision making, GMC, London (2002). 7 N.S. Jecker and R.A. Pearlman, Medical futility. Who decides?, Arch Intern Med 152 (1992) (6), pp. 11401144. Abstract + References in Scopus | Cited By in Scopus 8 C.M. Watts and W.A. Knaus, The case for using objective scoring systems to predict intensive care unit outcome, Crit Care Clin 10 (1994) (1), pp. 7389 [discussion 912]. 9 R.B. Becker and J.E. Zimmerman, ICU scoring systems allow prediction of patient outcomes and comparison of ICU performance, Crit Care Clin 12 (1996) (3), pp. 503514. Abstract | Abstract + References in Scopus | Cited By in Scopus 10 D.H. Beck, B.L. Taylor, B. Millar and G.B. Smith, Prediction of outcome from intensive care: a prospective cohort study comparing acute physiology and chronic health evaluation II and III prognostic systems in a United Kingdom intensive care unit, Crit Care Med 25 (1997) (1), pp. 915. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 11 P.S. Kamath, R.H. Wiesner, M. Malinchoc, W. Kremers, T.M. Therneau and C.L. Kosberg et al., A model to predict survival in patients with end-stage liver disease, Hepatology 33 (2001) (2), pp. 464470. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 12 L. Donaldson, On the state of public health: the annual report of the Chief Medical Officer, Department of Health, London (2001). 13 Mental Capacity Act 2005. HMSO. 14 S.M. White and T.J. Baldwin, The Mental Capacity Act 2005implications for anaesthesia and critical care, Anaesthesia 61 (2006) (4), pp. 381389. Full Text via CrossRef 15 Age Concern, October 2005. Advance statements, advance directives and living wills. 16 The European Convention on Human Rights 1998; Articles 2, 3 and 8. 17 T. Beauchamp and J. Childress, Principles of biomedical ethics (4th ed), Oxford University Press, Oxford (1994). 18 J. Rogers, S. Ridley, P. Chrispin, H. Scotton and D. Lloyd, Reliability of the next of kins estimates of critically ill patients quality of life, Anaesthesia 52 (1997) (12), pp. 11371143. Abstract + References in Scopus | Cited By in Scopus 19 Portsmouth NHS Trust vs. Wyatt & Ors, EWHC 2247, 2004 (Fam.). 20 British Medical Association, Withholding and withdrawing life-prolonging medical treatment, BMJ Books, London (1999). 21 J. Way, A.L. Back and J.R. Curtis, Withdrawing life support and resolution of conflict with families, BMJ 325 (2002) (7376), pp. 13421345. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 22 P. Levack, Live and let die? A structured approach to decision-making about resuscitation, Br J Anaesth 89 (2002) (5), pp. 683686. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 23 R.D. Troung and J.P. Burns, Excellence in end-of-life care: a goal for intensivists, Intensive Care Med 28 (2002) (9), pp. 11971199. 24 I.D. Nesbitt and D.W. Ryan, End of life care in ICU, Care of the Critically Ill 21 (2005), pp. 99100. Abstract + References in Scopus | Cited By in Scopus 25 M.D. Dowdy, C. Robertson and J.A. Bander, A study of proactive ethics consultation for critically and terminally ill patients with extended lengths of stay, Crit Care Med 26 (1998) (2), pp. 252259 [Erratum in: Crit Care Med 1998 Nov;26(11):1923]. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 26 The Ethox Centre. www.ethox.org.uk. 27 A.M. Slowther and T. Hope, Clinical ethics committees, BMJ 321 (2000) (7262), pp. 649650. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 28 B v An NHS Trust, EWHC 429, 2002 (Fam.). 29 C. Dyer, Woman makes legal history in right to die case, BMJ 324 (2002) (7338), p. 629. Full Text via CrossRef | Abstract + References in Scopus | Cited By in Scopus 30 An NHS Trust v A & Anor (adult patient: withdrawal of medical treatment) EWCA 1145, 2005 (Civ.). 31 An NHS Trust v MB & Anor, EWHC 507, 2006 (Fam.). Non-invasive for end of life (Crit Care Med 2008;36:789) From: Stephen Streat (ADHB) <StephenS@adhb.govt.nz> Date: Sun, May 10, 2009 at 10:56 PMSubject: Re: [CCM-L] persistent myoclonus To: Discussion of Critical Care Medicine <ccm-l@ccm-l.org>I’ve been off-line for a few days (conference in Sydney), but would like to respond to this because I think that there is one very neat treatment for this condition — which we use frequently, think is superior to everything else on several grounds and I can recommend — thiopentone infusion.Before you dismiss this post — let me explain …If this truly is post-anoxic status myoclonus, in a situation where there has been a profound severe hypoxic-ischaemic insult (e.g. prolonged arrest, delayed CPR etc etc), then as Michaal and others have pointed out, it is a VERY bad sign and forms a strong part of a decision to withdraw treatment. Of course it needs to be distinguished from transient intermittent seizures and from the Lance-Adams syndrome, which do not share the dire prognostic significance of status myoclonus in this context. But I’m assuming that someone has done that and that you are dealing with “the real thing”.First thing to say is “treating it with curative intent” is a strategy which is doomed to fail. Don’t try. All you will do is produce either a delayed death, or another Schiavo (see attached PDF, read the first few pages especially).Second thing — if status myoclonus is “bad” (as it often is) it is awful to look at — for both the healthcare workers and the patients family. Sometimes (I’ve seen it), it can cause heatstroke ! (in the patient). I’ve seen one patient recently (in whom I let it go on for 30 minutes to show a “blind” family what I was talking about) who developed a fever (42C), profound shock and a lactate of 20 mmol/l in just 30 minutes. I actually reversed all this (noradrenaline, 3 litres of iced-saline, thiopentone, external cooling) just to keep the patient alive for the next few hours, so that I could hold another couple of family meetings, including waiting for another family member to arrive by plane.Third thing — WHEN you have decided that “treatment with curative intent” is not appropriate, you then have the issue of “how do I palliate this awful phenomenon”. This is what thiopentone infusion does — it controls the phenomenon very well, without much of a “loading dose”, and with usually only a modest (150-250 mg/hr) infusion rate. It also does this with remarkable haemodynamic stability (you have probably already volume loaded the patient well, prior to the decision to provide symptomatic control only). It does not stop the patient from breathing (after all, the brain stem is usually fairly well preserved). So — you can (and we do) then wean the patient from IPPV to spontaneous breathing, and then extubate them and pull out all the lines (ETT, NG. art line, cease all monitoring) keeping only one line for thiopentone, and a Foley catheter ..The patient looks peaceful (and is), is devoid of NG and ETT (so their face looks “near-normal”) and there is no ventilator, ECG, art-line or pulse-oximetry — only the family, the nurse, conversation, weeping, singing, sometimes some music, a few drinks (yes reader, a few drinks), laughter and comfort — and no evident distress, from the patient, family or nurses/doctors. Meanwhile — thiopentone is cumulating all the time that myoclonus is being controlled. Typically the patients die a few hours after extubation.Typically I might chart “Thiopentone, 150mg/hr by infusion + 100mg IV prn for control of status myoclonus” and review the infusion rate every few hours. If the patient is getting a lot of prn, I might write “Thiopentone 300mg/hr plus 50mg IV prn.” Nurses are very happy with this. Cheap, effective monotherapy with good symptom control, allows for highly “low-tech” good comfort care and peaceful death. Very smooth. No panic moments. Comes recommended with 4 or 5 pale yellow stars. No RCTs. YMMD.Comments ?Perhaps I should write up a small case series of ten or so to raise peoples awareness of this option.
Treating Pneumonia in Patients with Dementia
abx increased survival at the expense of comfort (Arch Intern Med 2010;170(13):1102
Advanced Directives
defining advanced directives from chest
How you say it
Framing no CPR as the norm strongly influenced family Also using the alternative as “allow natural death” (Crit Care Med 2013;41:1686)
Benzos and Opioids do not hasten death
PMID 15249473
Give them a Brochure
N Engl J Med. 2007 Feb 1;356(5):469-78. A communication strategy and brochure for relatives of patients dying in the ICU.