Lacunar/small vessel strokes-usually eithe risolated sensory or isolated motor
Cortical/large vessel=alterations of cosciousness, thinking, language
Ant=leg, Arm/face=MCA, Homonymous hemianopsia=PCA
Must, must do sensory exam inthese patients, often times they will not complain fo sensory c/o
NNT
from Michael Liao
NINDSNNT 6 (42.7% vs 26.5%)NNH 17 (6.4% vs 0.6%)ECASSIIINNT 14 (52.4% vs 45.2%)NNH 47 (2.4% vs 0.2%)…. 23 (7.9% vs 3.5% with NINDS definitions)NNT is based on mRS <= 1 at 3 moNNH is based on symptomatic ICHI use all the NINDS data as it appears they skewed both part 1 andpart 2 (artificially(?)) to have approx half of the patients in the0-90min time windows. I find NINDS a bit trickier to piece back together than ECASS3.
—
Expansion of TPA window to 4.5 hours by AHA (Stroke 2009;40:)
ECASS III
Table 1 ECASS III Exclusion Criteria
- Age < 18 or > 80 years
- Onset of stroke > 4.5 hours before drug administration or symptom onset unknown
- Stroke symptoms present < 30 minutes or significantly improving before treatment
- Intracranial hemorrhage
- Severe stroke as defined by NIHSS > 25 or imaging (CT or MRI) displaying > 1/3 of middle cerebral artery territory involved
- Seizure at the onset of stroke
- Stroke or serious head trauma within the previous 3-months
- Combination of previous stroke and diabetes mellitus
- Heparin within the preceding 48 hours with PTT above normal limit
- Platelet count < 100,000 mm3
- Systolic blood pressure > 185 mm Hg or diastolic > 110 mm Hg or aggressive treatment (intravenous medication) to reduce blood pressure to these limits
- Glucose < 50 mg/dL or > 400 mg/dL
- Symptoms suggestive of subarachnoid hemorrhage even if CT normal
- Oral anticoagulation therapy
- Major surgery or severe trauma within 3-months
- Other major disorders with an increased risk of bleeding
The Evidence Thrombolysis with Alteplase 3 to 4.5 Hours after Acute Ischemic Stroke, N Engl J Med 2008; 359: 1317-1329. t-PA Placebo NNT NNHGood Recovery at 90 Days 52.4% 45.2% 14 N/A Symptomatic ICH 2.4% 0.3% N/A 47Death 7.7% 8.4% N/A N/A
Tenecteplase vs. Alteplase-New inclusion criteria, manufacturer supported, (N Engl J Med 2012;366:1099-107.)
Acute Stroke with NIHSS Score of 0
Physicians rely on the National Institutes of Health Stroke Scale (NIHSS) to evaluate patients with suspected acute stroke and to make decisions about acute treatment. The NIHSS correlates with infarct size, clinical severity, and long-term outcome. It is important to recognize, however, that ischemic stroke may cause symptoms that are not captured by the NIHSS scale.
The NIHSS scale is highly weighted toward deficits caused by anterior circulation strokes, whereas deficits caused by posterior circulation strokes receive fewer points (1-3). Within the anterior circulation, the scale underestimates the degree of right versus left hemisphere injury (1,4). It is possible that some patients with persistent symptoms on arrival to ED and an NIHSS score of 0 still have an infarct (1).
In a recent study, stroke patients with an NIHSS score of 0 most commonly presented with nausea, vomiting, and headache, all of which are associated with posterior circulation ischemia (1). Midline lesions of the cerebellum cause truncal ataxia, which is not part of the NIHSS. In addition, decreased visual acuity, Horner’s syndrome, and memory impairments are neurologic deficits not captured by the NIHSS. Subtle limb weakness (4/5) in an upper motor neuron pattern (extensors of the arms or flexors of the legs) may not be observed on the motor component of the NIHSS.
These data reinforce that the NIHSS cannot replace history and a thorough neurologic exam to diagnose acute stroke and that the NIHSS alone cannot be used to rule out a stroke in patients with acute persistent symptoms.
References:
(1) Martin-Schild S, et al. Zero on the NIHSS Does Not Equal the Absence of Stroke Ann Emerg Med 2010 Sep 8. [Epub ahead of print]
(2) Libman RB, et al. Differences between anterior and posterior circulation stroke in TOAST Cerebrovasc Dis 2001;11:311316.
(3) Sato S, et al. Baseline NIH stroke scale score predicting outcome in anterior and posterior circulation strokes Neurology 2008;70:23712377.
(4) Fink JN, et al. Is the association of National Institutes of Health Stroke Scale scores and acute magnetic resonance imaging stroke volume equal for patients with right- and left-hemisphere ischemic stroke? Stroke 2002;33:954958.
(Source Unknown Sorry!!!!!)
2009 AHA Stroke Imaging Recs
Acute Stroke, Neuroimaging, and Thrombolysis
- MRI is at least equal in efficacy to CT for detection of ICH in the hyperacute stroke patient, and both appear to have very high sensitivity and specificity. MRI is superior to CT for demonstration of subacute and chronic hemorrhage and hemorrhagic transformation of an acute ischemic stroke.
- MR-DWI (diffusion weighted imaging MR) is far superior to unenhanced CT and routine MRI in the detection of acute ischemia, with very high sensitivity and specificity. For a patient within 3 hours of symptom onset, MRI can be used if it does not unduly delay the timely administration of IV tPA since a more definitive diagnosis will be obtained with MR-DWI and it is far more effective than CT for excluding some mimics of acute cerebral ischemia.
- For patients beyond 3 hours from onset of symptoms, either MR-DWI or CTA should be performed, especially if mechanical thrombectomy or intra-arterial thrombolytic therapy is contemplated.
- Frank hypointensity on CT, particularly if it involves more than one third of an MCA territory, is a strong contraindication to treatment with thrombolysis. Early signs of infarct on CT, regardless of their extent, are not a contraindication to treatment.
- Gradient-echo MR can detect microhemorrhage, both old and new, better than CT, indicating the presence of amyloid angiopathy, hypertension, small vascular malformations, and other vascular diseases. The presence of a small number of these microhemorrhages (< 5) does not contraindicate intravenous thrombolysis.
Reference: Latchaw RE,et al. Recommendations for imaging of acute ischemic stroke: a scientific statement from the American Heart Association Stroke 2009;40(11):3646-78.
_____
Advanced Stroke Imaging References
Early CT Signs of Ischemic Stroke
from emedhome
The NINDS Study found a 31% sensitivity for early signs of ischemic stroke on noncontrast CT within 3 hours of symptom onset. The rate of detection increased to 82% at 6 hours (1).
Early signs of cerebral ischemia on CT:
- Hypoattenuation of brain tissue – with ischemia, cytotoxic edema develops resulting in increased brain water content. There is a loss of gray-white differentiation because of the increase in the relative water concentration within the ischemic tissues.
- With edema, swelling of the gyri produces sulcal effacement, which may lead to ventricular compression.
- Hyperdense MCA sign – a result of thrombus or embolus in the MCA.
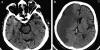
- Obscuration of the lentiform nucleus (also called blurred basal ganglia) is seen in MCA infarction and is one of the most frequently seen signs.
- Insular Ribbon sign refers to hypodensity and swelling of the insular cortex (the center of the cerebral cortex deep between the temporal lobe and the frontal lobe).
The sooner these signs become evident, the more profound is the degree of ischemia (1,2). Typically, at 6-12 hours sufficient edema is recruited into the stroke area to produce significant regional hypodensity on CT; a large hypodense area present within 3 hours of reported symptom onset should prompt careful review regarding the time of stroke symptom onset.
There is controversy as to whether early signs of infarct on CT are a contraindication to thrombolysis. The presence of CT evidence of infarction early in presentation has been associated with poor outcome and increased propensity for hemorrhagic transformation after thrombolytics in some studies (3,4). In the NINDS trial, there was no interaction between early infarction signs and tPA treatment for any clinical outcomes. Currently early signs of ischemia on CT are not generally considered to be a contraindication to use of tPA. However, “frank hypointensity” on CT, particularly if it involves more than one third of an MCA territory, is a strong contraindication to treatment (1).
References:
(1 ) Latchaw et al. Recommendations for imaging acute ischemic stroke: A scientific statement from the American Heart Association Stroke 2009;40:3646-78.
(2) Patel SC, et al. National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. Lack of clinical significance of early ischemic changes on computed tomography in acute stroke JAMA. 2001;286: 28302838.
(3) von Kummer R, et al. Acute stroke: usefulness of early CT findings before thrombolytic therapy Radiology1997;205(2):327-33.
(4) Dzialowski I, et al. Extent of early ischemic changes on computed tomography (CT) before thrombolysis: prognostic value of the Alberta Stroke Program Early CT Score in ECASS II Stroke 2006;37(4):973-8.
80-85% of strokes
majority are thrombotic vs. embolic (Mitral Stenosis, MI, A-Fib)
Hallmark of stroke is sudden onset of focal neurological derangement in a vascular area
Differential
Hemorrhage Migraine Hyperglycemia Carotid Dissection Todd’s Paralysis Bell’s Encephalitis/Abscess Temporal Arteritis Hypoglycemia Hyponatremia Hypertensive Encephalopathy Air Embolism Multiple Sclerosis Dementia
Common Major Stroke Syndromes
Anterior Circulation
Frontoparietal Lobes
Anterior Aspect of Temporal Lobes
Optic Nerve and Retina
Deep Gray Matter Structures
Posterior Circulation
Medial Aspect of Temporal Lobes
Visual Occipital Cortex
Thalamus
Brainstem
Upper Spinal Cord
Cerebellum
Auditory and Vestibular Aspects of the Ear
Anterior cerebral artery
Paralysis mainly of opposite leg and mild arm involvement
Sensory deficits paralleling paralysis
Altered mentation, confusion, judgment, and impaired insight
Gait apraxia (clumsiness)
Bowel and bladder incontinence
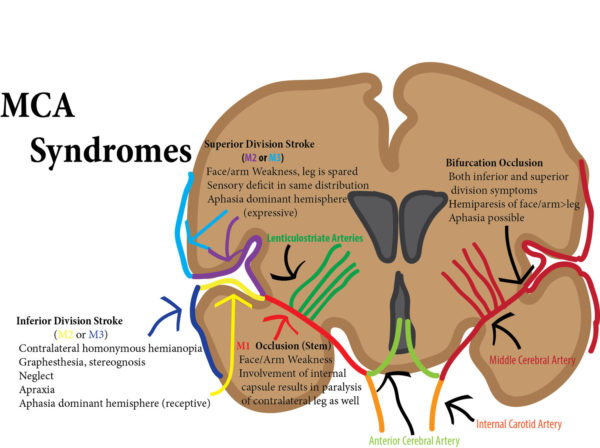
Middle cerebral artery
Paralysis of opposite side of body; arm and face worse than leg
Sensory deficits paralleling paralysis
Blindness in half of visual field (hemianopsia)
Aphasia (if dominant hemisphere involved, usually left)
Hemineglect (If non-dominant hemisphere, usually right)
Inability to recognize known objects (agnosia)
If Gaze preference, patients look towards the lesion
Posterior cerebral artery
Blindness in one half of visual field (hemianopsia)
Third nerve paralysis
Lack of visual recognition (visual agnosia)
Altered mental status with impaired memory
Cortical blindness
Brainstem/Cerebellum
Crossed Signs-face one side, body the other
Hemiparesis or quadriparesis (or worse yet locked in syndrome)
Sensory loss, hemi or all 4 extremities
Diplopia
Dysconjugate Gaze
Nystagmus
Dysarthria/Dysphagia
Vertigo
Decreased LOC or syncope
Ataxia
Vomitting
Patients presenting with pontine infarction may describe a preceding transient pain radiating from the unilateral eye to the nose, following which they developed numbness or ataxic hemiparesis on the side contralateral to the pain.4 The beauty parlor syndrome has been described in elderly patrons receiving shampoo treatments. Mechanical impingement by neck rotation and hyperextension decreases vertebral artery flow and produces hypoperfusion at the atlanto-occipital-distal vertebral artery junction. Patients may present with vertigo and ataxia.5
Evaluation
Stroke notification
Check Glucose
Consider Aortic Dissection or if neck pain in the absence of trauma, consider arterial dissection of neck vessels
Obtain a BP in both arms
NIH Stroke Scale (FERNE) , but remember it leaves out some CN, gait, and nystagmus
EKG
Draw Labs-CBC, PT/PTT, Lytes, C-XR, Consider ABG, C-Spine, LFTs
Get a stat CT Minus Head
multimodal MRI including gradient echo is at least as and probably more sensitive for bleeds than CT (J Neuro Neurosurg Psych 2001 Apr;70 suppl 1:I7-11)

Management
BP
One study showed cardene/labetalol lowering of BP before TPA may be assoc. with higher adverse events, but it may just be that patients with HTN do worse (Arch Neurol 2008;65:1174)
Keep MAP<130
Positioning
Hob flat may increase CPP (Neurology 2005;64:1354)
Cerebral Edema
Elevate head of bed to 30º
Hyperventilation or Mannitol only if acute deterioration
Seizures
Only to prevent recurrent seizures
Anticoagulation
ASA well proven (huge Chinese study)
No harm if you give it in hemorrhagic stroke, but still not advised (Stroke, 2000;31:1240-1249)
No benefit to other anticoagulants unless concerns over pt with current A.Fib (Stroke 33:856, 2002)
TPA
lytics up to 4.5 hours (NEJM 2008;359(13):1317)
Must be read by neuroradiologist (Neurologists, general radiologists, and certainly ER docs are not qualified)
BP must be less than 185/110
The NINDS trial was divided into two parts; in both parts, patients were randomized to receive tPA, 0.9 mg/kg, or placebo within three hours after onset of stroke symptoms. Patients in the treatment arm were given 0.9 mg/kg tPA (maximum dose, 90 mg) in a 10% bolus followed by a constant infusion of the remaining 90% over 60 minutes. The first part enrolled 291 patients and measured whether they had improvement in their NIHSS score of four points or more over baseline or resolution of deficits within 24 hours of onset of stroke. In this portion of the trial, there was no statistical difference between the tPA group and the placebo recipients (P = 0.21). However, in post hoc analysis, the authors point out that the median NIHSS scores were two points lower in the 0- to 90-minute range and four points lower in the 90- to 180-minute range when the tPA group was compared with placebo (P value not provided). The second part enrolled 333 patients and assessed clinical outcomes at three months. Patients who received tPA were at least 30% more likely to have minimal or no disability based on four measures of neurologic disability (NIHSS, modified Rankin, Glasgow outcome scale, and Barthel Index). The primary hypothesis in part two was tested with a global statistic to simultaneously test for effect in all four outcome measures. Symptomatic intracerebral hemorrhage occurred in 6.4% of tPA recipients but only 0.6% of placebo recipients (P < 0.001). Mortality at three months was 17% in the tPA group and 21% in the placebo group (P = 0.30). A number-needed-to-treat analysis of the NINDS trial indicates that for every eight acute stroke patients treated, one will benefit. One patient in 17 will suffer an intracranial hemorrhage, and one in 40 will die. These numbers may be helpful in explaining risks and benefits to patients who are eligible for thrombolytic treatment.
Meta-analysis of current thrombolytic for stroke data Symptomatic ICH 6.2% Absolute Risk Increase Fatal ICH 2.5% Absolute Risk Increase (Stroke. 2003 Jun;34(6):1437-42.)
The major risk of t-PA is symptomatic intracerebral hemorrhage, which occurred in 6.4 percent of patients who received t-PA, as compared with 0.6 percent of patients who received placebo.1 These figures represent an absolute difference in the risk of symptomatic intracerebral hemorrhage of 6 percent. Seventy-five percent of the patients with a symptomatic intracerebral hemorrhage were dead at three months. Yet despite the risk of intracerebral hemorrhage, the mortality at three months was insignificantly lower in patients treated with t-PA (17 percent) than in placebo-treated patients (21 percent). Two reasons may underlie the similar mortality of t-PAtreated and placebo-treated patients despite a higher risk of symptomatic intracerebral hemorrhage among patients treated with t-PA. First, most patients who had an intracerebral hemorrhage had large strokes and were likely to do poorly regardless of the presence of intracerebral hemorrhage within the damaged brain. Secondly, t-PA probably makes some “big” strokes much smaller. The patients with small strokes are less likely to die. Patients treated with t-PA who have very large strokes (very severe neurologic deficits, meaning an NIH stroke scale score greater than 20) and who already have evidence of a large acute ischemic stroke on baseline CT have an increased risk of symptomatic intracerebral hemorrhage. 17 However, patients in these two subgroups who receive t-PA are more likely to return to normal than patients who are treated with placebo. The decision to use t-PA in these patients should be made only after frank discussion of the potential risks and benefits with both the patient and the family. Figure 1 adapted from Marx J. Classification system for stroke patients. Proceedings of the National Symposium on Rapid Identification and Treatment of Acute Stroke, December 13, 1996. Washington, D.C.: National Institute of Neurological Disorders and Stroke (NINDS), November 1997.
Contraindications to TPA
Get TEE, eval ekg for A-Fib. Carotid dopplers.
ASA or plavix
Site for NINDS Group Guidelines for TPA
editorial on why it has not been adopted more widely (Lancet 2006;5:722)
ECASS-III
Endovascular Treatments
From Resus.me by Cliff Reid:
Two randomised controlled trials have been published which compare endovascular stroke treatments with intravenous tPA. Both the American Interventional Management of Stroke (IMS) III trial (1) and the Italian SYNTHESIS Expansion trial (2) had Modified Rankin Scores as their primary endpoint. No significant differences in this outcome or in mortality or intracranial haemorrhage rates were found in either trial, and IMS III was terminated early due to futility.
A third trial, from North America, called MR RESCUE, randomised patients within 8 hours after the onset of large vessel, anterior-circulation strokes to undergo mechanical embolectomy or receive standard care(3). No clinical outcome differences were demonstrated.
An accompanying editorial (4) draws the following conclusion:
“The IMS III and SYNTHESIS Expansion studies show that intravenous thrombolysis should continue to be the first-line treatment for patients with acute ischemic stroke within 4.5 hours after stroke onset, even if imaging shows an occluded major intracranial artery. Beyond 4.5 hours, the MR RESCUE trial does not provide data supporting the use of endovascular treatment in patients with an ischemic penumbra of any size.”
Many might argue that showing endovascular treatment is equivalent to thrombolysis just means endovascular treatment doesn’t work, because a significant proportion of the emergency medicine community views this as the correct interpretation of a thorough analysis of the stroke thrombolysis literature.
1. Endovascular Therapy after Intravenous t-PA versus t-PA Alone for Stroke
NEJM Feb 8, 2013 Full Text Link
2. Endovascular Treatment for Acute Ischemic Stroke
NEJM Feb 8, 2013 Full Text Link
3. A Trial of Imaging Selection and Endovascular Treatment for Ischemic Stroke
NEJM Feb 8, 2013 Full Text Link
4.Endovascular Treatment for Acute Ischemic Stroke — Still Unproven
NEJM Feb 8, 2013 Full Text Link
Endovascular Therapy after Intravenous t-PA versus t-PA Alone for Stroke J.P. Broderick and Others | N Engl J Med 2013;368:893-903 | No benefit when IA was added to IV tPA
Cerebellar Stroke
Neurosurgery consult
Misc.
Elevated CK-MB after stroke are not necessarily indicative of AMI, need elevated Troponin to properly eval. (Stroke 33:286 2002)
Giving ASA even to hemorrhagic stroke did not cause additional mortality or increased bleeds (Stroke 2000; 31:1240-9)
Old Stroke Patients
may still be safe to lyse patients > 80 y/o (BMJ 2010;341:c6046)
Andy has editorial as well (ACEP News Feb 2011)
Young Stroke Patients
Strokes in Younger Patients (EMEDhome)
When evaluating a younger patient in the Emergency Department with a stroke, keep in mind the following:
- Cardiac causes including Right-to-Left shunts from a patent foramen ovale (PFO) or occult atrial septal defect (ASD).
- Cocaine and amphetamine use
- Atrial myxoma
- Cervical artery dissections. Approximately 20% of strokes in the young are caused by carotid artery and vertebral artery dissections in the neck, compared to 2.5% in older patients.
As transesophageal echocardiography (TEE) is considered the most sensitive method to date to detect PFO (1), think to suggest a TEE as part of the evaluation.
“In the absence of other causative conditions, an ASD or PFO may be presumed to be the underlying cause of cerebrovascular thromboembolism. The search for these defects will be more cost-effective in younger stroke patients who, unlike older patients, rarely have the cardiovascular conditions associated with advanced age that commonly cause strokes in the elderly” (2).References: (1) Horton SC, Bunch TJ. Patent foramen ovale and stroke Mayo Clin Proc 2004;79(1): 79-88. (2) Jaber WA, et al. Suspect an atrial septal defect if a young patient has a stroke Cleveland Clin J Med 2001; 68: 954-956. (3) Cabanes L, et al. Atrial septal aneurysm and patent foramen ovale as risk factors for cryptogenic stroke in patients less than 55 years of age. A study using transesophageal echocardiography. Stroke 1993; 24:1865-1873. (4) Jones J, Geninatti M. Cardiology Emerg Med Clin North Am 1997;15(2):341-63.
from emedhome.com
Think Endocarditis in Young Patients With Apparent Stroke Neurologic complications occur in approximately one-third of cases of infective endocarditis and typically present as a stroke syndrome.
Valvular vegetations can embolize to the brain and causes cerebral infarction, usually in a branch of the middle cerebral artery, causing contralateral motor or sensory symptoms. However, infarction can occur anywhere in the cerebral circulation, producing a myriad of possible neurologic symptoms. Infectious vegetations can embolize to branch points in the cerebral vascular tree, causing the formation of mycotic aneurysms. A mycotic aneurysm is an aneurysm that results from an infectious process that involves the arterial wall. These are noted in up to 2% of patients with infective endocarditis, causing symptoms due to mass effect, vascular leakage, or sudden rupture. This can lead to a broad spectrum of clinical presentations ranging from subarachnoid hemorrhage to meningitis to coma.
Infective endocarditis is therefore an important consideration when a young person who has no risk factors for premature cerebrovascular disease who presents to the ED with stroke symptoms, particularly when the patient has risk factors for the development of infective endocarditis, such as IV drug use or valvular heart disease, or presents with a fever. References: (1) Mattu A, Goyal D. Emergency Medicine: Avoiding the Pitfalls and Improving the Outcomes © Blackwell, 2007 (2) Crawford MH, Durack DT. Clinical presentation of infective endocarditis Cardiol Clin 2003;21: 159-66. (3) Tunkel AR, Kaye D. Neurologic complications of infective endocarditis Neurol Clin 1993;11: 419-40.
Exam
(Jama 2005;293(19):2391)
LR of the absence of facial paresis, arm drift, and abnormal speech is 0.39
Oxfordshire Classification of Subtypes of Cerebral Infarction*
Totalanterior circulation infarction syndrome (TACS)
-
- A combinationof new higher cerebral dysfunction (ie, dysphasia, dyscalculia, visuospatial disorder); homonymous visual field defect; and ipsilateral motor and/or sensory deficit of at least 2 areasof the face, arm, and leg.
Partial anterior circulation infarction syndrome (PACS)
-
- Only 2 of the 3 components of theTACS syndrome are present, with higher cerebral dysfunction alone, or with a motor/sensory deficit more restricted thanthose classified as LACS (ie, confined to 1 limb or to faceand hand, but not to the whole arm).
Lacunar infarctionsyndrome (LACS)
-
- Pure motor stroke, pure sensory stroke, sensori-motorstroke, or ataxic hemiparesis.
Posterior circulation infarction syndrome (POCS)
-
- Any of the following: ipsilateral cranial nervepalsy with contralateral motor and/or sensory deficit; bilateralmotor and/or sensory deficit; disorder of conjugate eye movement; cerebellar dysfunction without ipsilateral long-tract deficit(ie, ataxic hemiparesis); or isolated homonymous visual fielddefect.
*Based on data from Bamford et al.54
Sub-Arachnoid Hemorrhage (SAH)
Posterior Circulation Stroke
Neurologists have described acute confusional states and acute agitated delirium with several sites of infarction [4, 5 and 6]. Some have been bilateral, others on the right (non-dominant) or the left (dominant) specifically. Restlessness, agitation and disorientation are common with posterior artery infarctions. Forced crying out, cursing and unintelligible speech often have been reported in these cases, as they were in our patient [2]. Hypertension and generalized slowing on EEG are described in some patients, whereas others have normal CT scans for some time before changes of density can be identified [2]. The course of delirium can last several weeks with varying outcomes. Older age, comorbidity, and extent of the infarction have significant impact. Our patient had a history that included diabetes mellitus, hypertension, adenocarcinoma, and psychiatric disorders, all of which could have produced some of the initial symptoms. It is unlikely that her fever was due to medications, because she had been taking them for weeks, or that it was due to infection, because an organism could not be cultured and the white blood cell count was persistently normal. The maximum outside temperature on that day was 76°C.
The Amitriptyline and Nortriptyline levels were less than 10 ng/ml. It is known that acute strokes can cause elevated temperature, agitation and hypothermia. Although total CK level was not obtained, it is unlikely that agitation caused the fever, as it persisted even after agitation resolved with sedation. The ultimate cause of her fever was puzzling, but the most likely etiology was the stroke. Whether or not the antibiotics prescribed at the onset eradicated an undetected infection was never resolved. The confirmed diagnosis of left posterior cerebral artery infarction certainly could have explained the majority of her signs and symptoms as well as the course.
It is quite important to consider this diagnosis early as it carries a high mortality, particularly in the elderly [2]. Rapid intervention with TPA is increasingly initiated to spare neuronal damage when it can be administered shortly after an event. It would not have been given to this patient 11 h post infarction. Because initial CT scans may not be diagnostic, an MRI or MRA is more definitive. Plan for workup depends on initial clinical evaluation, although in most hospitals the first imaging study would be a CT scan. A CT scan can exclude unexpected large lesions or the presence of bleeding but other investigations are necessary to identify the stenotic or occluded artery. Abnormal diffusion weighted imaging (DWI) is a very sensitive and specific identifier of ischemic stroke in patients presenting within 6 h of stroke. Although DWI can detect areas of ischemia within 1530 min of onset after only 3 s of imaging, its availability is limited. CT scans are more available, easily obtained and cheaper than MRIs or DWIs, so they are usually ordered first. But when non-diagnostic and at the earliest suspicion of ischemic stroke, the alternatives should be considered. Once the delirium clears, post-stroke depression should be reassessed. Untreated depression has a negative impact on both rehabilitation and long-term prognosis, and a premorbid history for depression significantly increases the risk for recurrence. Post-stroke depression is a well-known phenomenon and consistent with our patient’s symptoms [7]. When the anterior limbic system is disconnected from the destroyed posterior, the amygdala and anterior hippocampus can also induce confusion, fever, anger and amnesia. The infarct will also disrupt connections between the temporal neocortex and the limbic system, making it extremely difficult to use language-encoded memories. As time progresses to allow for collateral circulation, the symptoms may resolve if the infarction is not so large that it causes devastating destruction.
In conclusion, infarction of the posterior cerebral artery, whether unilateral or bilateral, may present first as delirium without many focal findings. When suspected, an MRI should be immediately obtained to help clarify a diagnosis. Intervention with TPA in all non-hemorrhagic strokes remains controversial but patients treated within 90 min definitely have improved outcomes. Early identification of the patients who may benefit from this approach is essential [8]. The affected limbic structures can produce early psychiatric symptoms involving delirium and altered affect and the later development of post-stroke depression.
ATLANTIS trial: Mild to moderate strokes 79% completely recover at 90 days vs. 56% for placebo. In moderate to severe, 33% TPA vs. 5% placebo. 22% change of intracranial hemorrhage. All symptomatic hemorrhages were fatal. (Stroke 2002, 33:493-496)
ANTIPLATELET DRUG DISCONTINUATION IS A RISK FACTOR FOR ISCHEMIC STROKESibon, I., et al, Neurology 62:1187, April 2004
BACKGROUND: Prophylaxis with antiplatelet drugs is effective for the secondary prevention of cardio- and cerebrovascular disease. Only limited information is available concerning the occurrence of ischemic stroke in the setting of discontinuation of such treatment.
METHODS: These French authors interviewed 320 patients (or surrogates) hospitalized for a transient ischemic attack (TIA) or stroke, regarding their use of an anti- platelet drug (almost always aspirin), and any changes in that treatment, in the month before hospitalization.
RESULTS: Of the minority (31, 9.7%) with a hemorrhagic stroke, none had discontinued an antiplatelet drug during the previous month, while six were being anticoagulated. Of 289 patients with an ischemic infarction, antiplatelet drug therapy was discontinued during the month before the episode in 13/103 who had been receiving it. The interval between discontinuation of anticoagulation and the occurrence of stroke was always 6-10 days, which is consistent with the timing of aspirin’s effect on platelets. In seven of the 13 cases, discontinuation of treatment was ordered by a physician prior to surgery, while in six it occurred due to “negligence” or based on the belief that treatment was not clinically relevant.
CONCLUSIONS: There may be an increased risk of stroke fairly shortly after discontinuation of antiplatelet drug prophylaxis. 8 references (igor.sibon@chu-bordeaux.fr)
NINDS study: inclusion criteriaischemic stroke with clearly defined time of onset; measurable deficit on National Institutes of Health (NIH) stroke scale; no evidence of intracranial hemorrhage on computed tomography (CT) of brain; treatmentt- PA 0.9 mg/kg for maximum of 90 mg (10% given as bolus, 90% as constant infusion over 60 min [smaller than cardiac dose]); patient not to receive anticoagulants or antiplatelet drugs for 24 hr after treatment; strict protocol for blood pressure monitoring and control; exclusion criteriaprior stroke or head trauma within 3 mo; major surgery in previous 14 days; history of intracranial hemorrhage; hypertension; suspected transient ischemic attack (TIA); hypothesisconsistent and persuasive difference between t-PA and placebo in patients who recover with minimal or no deficit 3 mo after treatment; outcomet-PA group had higher percentage of favorable outcomes; no increase in severe disability or death resulting from administration of t-PA; however, you were ten times more likely to develop a symptomatic intracranial hemorrhage resulting from your stroke if you got t-PA than if you didnt; mortality from symptomatic intracranial hemorrhage 45% higher than mortality from disease process itself; t-PA patients 32% to 55% more likely to have complete recovery from stroke than placebo group (11%-13% more patients have complete recovery with t-PA than without it); must treat 8 to 9 patients to obtain one additional complete recovery; all patients with ischemic stroke benefited equally, regardless of size; these results held at 6 mo and 1 yr Recommendations of Stroke Council of American Heart Association: published in 1996; t-PA should be given according to NINDS protocol by physicians with expertise in diagnosis of stroke and interpretation of CT; streptokinase should not be used; do not treat if CT signs suggest major infarct or treating facility cannot handle complications of intracranial hemorrhage (need neurosurgery backup plan); use caution in severe strokes (score >22 on NIH stroke scale); obtain informed consent
NIH stroke scale: fairly reliable and reproducible; primarily identifies lateralizing findings; evaluates 11 criteria; takes 5 to 8 min to perform; patient must have score >4 but <22 CT criteria: must be done within 3 hr of symptom onset; parenchymal hypodensity indicates large stroke or long interval between time of stroke and patients arrival; study showed correct interpretation of CT by emergency physicians in 66% of cases, 83% for neurologists and radiologists; only 70% of emergency physicians interpreted CT correctly 100% of time, 40% of neurologists and 50% of radiologists
Comparison of MRI and CT for Detection of Acute Intracerebral Hemorrhage
JAMA. 2004;292:1823-1830. Results The study was stopped early, after 200 patients were enrolled, when it became apparent at the time of an unplanned interim analysis that MRI was detecting cases of hemorrhagic transformation not detected by CT. For the diagnosis of any hemorrhage, MRI was positive in 71 patients with CT positive in 29 (P<.001). For the diagnosis of acute hemorrhage, MRI and CT were equivalent (96% concordance). Acute hemorrhage was diagnosed in 25 patients on both MRI and CT. In 4 other patients, acute hemorrhage was present on MRI but not on the corresponding CTeach of these 4 cases was interpreted as hemorrhagic transformation of an ischemic infarct. In 3 patients, regions interpreted as acute hemorrhage on CT were interpreted as chronic hemorrhage on MRI. In 1 patient, subarachnoid hemorrhage was diagnosed on CT but not on MRI. In 49 patients, chronic hemorrhage, most often microbleeds, was visualized on MRI but not on CT.
Prognosis and Decision Making in Severe Stroke
JAMA 2005;294(6):725
Our analysis suggests that EICs are prevalent within 3 hours of stroke onset and correlate with stroke severity. However, EICs are not independently associated with increased risk of adverse outcome after rt-PA treatment. Patients treated with rt-PA did better whether or not they had EICs, suggesting that EICs on CT scan are not critical to the decision to treat otherwise eligible patients with rt-PA within 3 hours of stroke onset. (JAMA. 2001 Dec 12;286(22):2830-8.)
Optimal stroke scale to lyse 10-20
Dizziness and Stroke
Stroke Among Patients With Dizziness, Vertigo, and Imbalance in the Emergency Department: A Population-Based Study Kerber KA, Brown DL, Lisabeth LD, et al. Stroke 2006;37:24847 Catherine Ambrose MDa aDenver Health Medical Center, Denver, Colorado Available online 30 March 2007. As part of the Brain Attack Surveillance in Corpus Christi (BASIC) project, this study sought to determine the percentage of stroke patients who presented to the Emergency Department (ED) with the complaints of dizziness, vertigo, or imbalance. This large population-based study examined all patients over the age of 44 years who presented to the ED or were directly admitted to the hospital between January 2000 and June 2003 with the previously mentioned isolated dizziness symptoms (DS) in Nueces County, Texas. The association of age, sex, race/ethnicity, and isolated dizziness symptoms with stroke or transient ischemic attack (TIA) was then determined using multivariable logistic regression. A total of 1666 patients were included in the study, with 3.2% (53 of 1666) ultimately diagnosed with stroke/TIA. Of those diagnosed, 23 presented with dizziness as the chief complaint, 18 with vertigo, 11 with imbalance, and 1 with more than one of the above symptoms. Isolated DS without additional neurological findings was a strong negative predictor of stroke/TIA (odds ratio [OR] 0.05; 95% confidence interval [CI] 0.020.11), whereas male sex was associated with an increased association with stroke/TIA (OR 2.5; 95% CI 1.44.4). Patients diagnosed with stroke/TIA were also found to be significantly older (69.3 ± 11.7 vs. 65.3 ± 12.9, p = 0.02). No significant difference in race/ethnicity was found between the stroke and non-stroke groups. When compared to dizziness, patients with imbalance were found to have an increased risk of stroke/TIA (OR 3.7; 95% CI 1.310.7), whereas no increased risk was found between those with vertigo versus dizziness (OR 0.9; 95% CI 0.42.0). The authors acknowledge several limitations to this study, including lack of evaluation of the majority of study patients in the ED by a neurologist, lack of magnetic resonance imaging on most study patients leading to possible undiagnosed strokes, and lack of comparison of symptom onset, duration, aggravating/alleviating factors, headache, and auditory symptoms between groups. The authors conclude that dizziness and vertigo are not associated with stroke/TIA, whereas patients who present with imbalance, those with additional neurologic findings, male gender, and older age are at the highest risk for possible stroke/TIA.
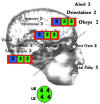
Rapid approach to NIHSS
The NIHSS as formulated by the National Institute of Neurological Disorders and Stroke has been applied at our medical center as the SQS and is shown here with maximal deficit scores clustered into 3 groups having a total of 7points each (Fig. 1). The level of consciousness section to the NIHSS is grouped together in the frontal regions with an aggregate deficit score totaling 7 for deficits in alertness, ability to answer questions, and obey commands. A second cluster of 3 consisted of deficits in sensation, extinction, and language being clustered along the motor strip and temporal-parietal regions; the extinction category tests inattention with complete neglect scoring 2. In regards to sensory deficits, this can be tested as withdrawal to noxious stimuli, with a severe loss scoring 2 points. A third cluster was placed in the posterior regions and included the maximal visual deficit score of 3 for bilateral hemianopia, and the near to unintelligible deficit score of 2 for dysarthria was placed alongside the maximal limb ataxia score of 2 in the cerebellar region; although speech dysarthria can be a result of ischemic damage to the motor strip, an alternative possibility would be dysarthria of a cerebellar origin and for convenience is grouped here in the posterior fossa region. The maximal motor deficit score of 4 for each of 4 extremities is grouped together (green circle, Fig. 1). Miscellaneous items that are not part of the 322 groupings are separated by a line parallel to the catho-metal line: best gaze (forced deviation of the eyes scores 2 points) and facial palsy (complete facial palsy scores 3 points). 3. Discussion Although not yet formally tested under controlled conditions, the SQS diagram may accelerate the time factor in reaching an accurate score, but formal testing of this hypothesis is needed. Although the NIHSS attempts to comprehensively include all regions of the brain that could be affected by stroke, the weighting factors for each functional item might need reassessment. The current equal weighting for arms vs legs remains reasonable for now, because 1 study showed very good reliability for these items with the 15-item NIHSS [6]. However, the same study found the least reliable items to facial palsy and dysarthria with low intraclass correlation coefficients being less than 0.40, and advocated a modified 11-item version of the NIHSS. In summary, the presented SQS diagram shown in Fig. 1, or similar versions, may facilitate training of examiners to comprehend the overall structure of the NIHSS system. The SQS diagram is intended only to serve as a supplement and not a replacement for current methods of scoring. However, further testing of a single visual composite for the NIHSS as an ancillary tool is needed to determine its actual usefulness. (AJEM 2008;26:189)
From EP Monthly:The years top stroke advances by Bobby Desai, MD
Lacunes & Lacunar Infarcts
The terms “lacune”, “lacunar infarct” and “lacunar stroke” are often used interchangeably, but they are not the same thing. Lacunes are 3 to 15 mm cerebrospinal fluid (CSF)-filled cavities in the basal ganglia or white matter, frequently observed coincidentally on imaging in older people, often not clearly associated with discrete neurological symptoms. “Lacunar stroke” describes a clinical stroke syndrome with the typical symptoms and signs referable to a small subcortical or brain stem lesion.1,2 “Lacunar infarct” should refer to a clinical stroke syndrome of lacunar type where the underlying lesion is an infarct on brain-imaging. On CT or MR T2-weighted and fluid-attenuated inversion recovery (FLAIR) imaging, an acute lacunar infarct can look just like a white matter lesion (WML), difficult to distinguish from an asymptomatic WML without diffusion-imaging to show a hyperintense signal (reduced on ADC), or a prior scan for comparison, especially in patients with WMLs. Some clinically evident acute lacunar infarcts may evolve with time into lacunes. These points are well-established.
Less well-established is how many clinically evident lacunar infarcts ever cavitate to become “lacunes”. It seems generally assumed that all lacunes start life as an infarct, even if the patient did not notice anything, and therefore share the same risk factors, etiology, prognosis, pathogenesis, etc, as clinically evident lacunar infarcts.35 However, suppose only a proportion of lacunar stroke lesions, perhaps as few as a third, ever cavitate, with the majority that fail to cavitate retaining the appearance of a WML?6 Counting lacunes could result in spurious risk factor and etiologic associations for lacunar stroke. We should not assume that the pathogenesis of clinically evident lacunar stroke is the same as for clinically silent lacunes. Equally, similarity in appearance between WMLs and clinically evident acute lacunar infarct could imply similar causation. However, surely the fact that one has caused symptoms (lacunar stroke/infarct) and the other not (WML/lacune) is important in itself and should lead to their careful distinction in any research at least until we know more.
Hemicraniectomy for Malignant Middle Cerebral
Bleck:
Perhaps I’m missing something, but as I read the case the patient was being treated with enoxaparin for secondary stroke prevention, presumably from a cardiac source since there would not be an indication to do so without a suspected cardiac source (excepting hospitals with the initials Must Give Heparin). Many stroke neurologists would think that four days is too early to start anticoagulation because of the risk of hemorrhagic transformation. This is an area of considerable controversy and little data, but a two week delay is commonly suggested. I didn’t think this should go by without comment. Now for a statement that may label me as a heretic, or an old guy to be ignored: I don’t think that the ease of use of enoxaparin justifies its use in this circumstance. I only use unfractionated heparin by intravenous infusion without a bolus in this setting, and keep the PTT about 70 sec. I don’t want a drug that may be too effective as an anticoagulant, or that I can’t reverse quickly. And I would not send a patient home this quickly when starting anticoagulation. Sorry, but this case demonstrates why.
Regarding treatments to stabilize the patient in order to get to the OR (or theatre for some of you, or operating department for people who object to the term room), this is one of the few circumstances in which more than modest hyperventilation is reasonable in the setting of ischemia; I might even hyperventilate more than David suggests, into the low to mid 20s for a brief period in order to get the patient to the OR. Regarding the mannitol dose, no one knows this either, but I’d probably start with 1 gm/kg on the theory that I want a high osmotic gradient and am not worried about rebound since the surgeon will make room for more swelling later. In recent years I have used a lot more hypertonic saline, but I realize that the literature is mixed in this regard; this would be a circumstance in which 30 – 60 mL of 23.4% saline (sodium quadrate in pharmacy parlance) over 10 min might be best because it seems to work most quickly. Coplin, what do you think as the holder of the IND for salt? Raise the head of the bed and give neuromuscular junction blockade so there is no coughing (boy am I old-fashioned!); there’s no point in ‘following the exam’ any longer. Turn the FiO2 up to 1.0 try to get some oxygen in without having to raise the blood pressure pharmacologically, although it is probably going up pretty high on its own (and if it isn’t, or is falling, the game is probably over already). Although there is no question that lowering core temperature lowers ICP, there isn’t time to do it here; the patient needs to be decompressed before any modality would work. I suppose I might start running in some chilled saline, which we keep around for post-cardiac arrest hypothermia, on the way to the OR, but I wouldn’t do anything that would delay transit.
I think the real question is whether to put in an external ventricular drain while the OR is being prepared. There is likely to be obstructive hydrocephalus here, and one might lower the ICP substantially by relieving it. The downside of this is the risk of upward herniation, which I have seen enough times to believe in and fear. Some folks will put one in prophylactically and clamp it to have it in place already when the patient’s exam deteriorates. If one does place an EVD, it shouldn’t be allowed to drain to atmospheric pressure; just take off a small amount of fluid. Since in the emergent situation the fluid will initially gush out, this is another argument for placing it before the patient is trying to die.
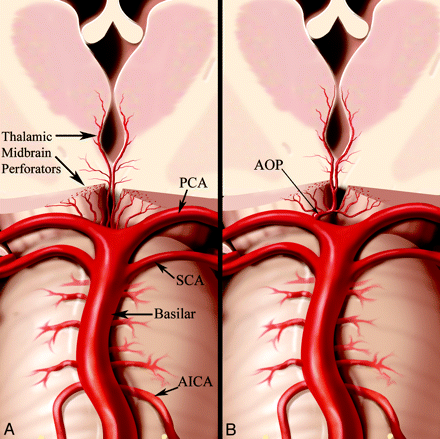
Basilar Artery Occlusion (BAO)
Administered IV Tpa up to 24 hours (48 hours if fluctuating) (Stroke 2011;42:2175)
TPA in patients on Coumadin
retrospective study showed increase post-tpa bleed rate even with INR < 1.7 (Arch Neurol 2010;67(5): Prabhakaran)
Critical Review Of Decomp Crani for Middle Cerebral Infarct
The Neurologist Issue: Volume 17(1), January 2011, p 6366
Posterior circulation stroke
Ischaemic stroke of the posterior circulation may cause mild or non-specific acute symptoms that are not captured on the National Institutes of Health Stroke Scale (NIHSS). A study published in the Annals of Emergency Medicine (2011;57:425) reveaed that patients with an NIHSS score of 0 and a documented infarct on diffusion-weighted imaging, frequently presented with headache, vertigo, nausea and truncal ataxia. This highlights the need to be alert to this pattern of symptoms in order to investigate further and rule out a posterior circulation stroke.
Neutrophilia
Stroke. 2008 Feb;39(2):355-60. Epub 2007 Dec 27.Early neutrophilia is associated with volume of ischemic tissue in acute stroke.
Good Care for Post-Stroke is most Important Factor
From EM Lit of Note Blog:
This is a prospective interventional study in which acute stroke units
in New South Wales Australia were randomized to either no protocolized
intervention, or an intervention with nursing protocols named above. At
the end of the three-year intervention period, 42% of the control group
had mRS 0 or 1 at 90 days, and 58% of the intervention group had mRS 0
or 1 at 90 days. There were small differences in the type of stroke,
education level, and prior ability to work that probably favored the
intervention group, but the differences at baseline were far smaller
than the magnitude of the treatment effect. In short, a basic nursing
protocol intervention improved outcomes more than any other intervention
for acute stroke.
“Implementation of evidence-based treatment protocols to manage fever,
hyperglycemia, and swallowing dysfunction in acute stroke (QASC): a
cluster randomised controlled trial.”
(Lancet, Volume 378, Issue 9804, Pages 1699 – 1706, 12 November 2011)
Crossed Leg Sign
Leg crossing is an easily obtained clinical sign and is independent of additional technical
examinations. Leg crossing within the first 15 days after severe stroke indicates a favorable
outcome which includes less neurologic deficits, better independence in daily life, and lower rates
of death. Neurology® 2011;77:1453–1456
Warfarin and tPA
tPA may be ok if the patient is taking warfarin but is sub-therapeutic (JAMA. 2012;307(24):2600-2608)
Neuroimaging Negative Stroke
Probably doesn’t exist as per EM Lit of Note
“Negative Diffusion-Weighted Imaging After Intravenous Tissue-Type Plasminogen Activator Is Rare and Unlikely to Indicate Averted Infarction” http://www.ncbi.nlm.nih.gov/pubmed/23572476
Speed to tPA
Time to Treatment With Intravenous Tissue Plasminogen Activator and Outcome From Acute Ischemic Stroke Jeffrey L. Saver, MD; Gregg C. Fonarow, MD; Eric E. Smith, MD, MPH; Mathew J. Reeves, PhD; Maria V. Grau-Sepulveda, MD, MPH; Wenqin Pan, PhD; DaiWai M. Olson, PhD; Adrian F. Hernandez, MD, MHS; Eric D. Peterson, MD, MPH; Lee H. Schwamm, MD JAMA. 2013;309(23):2480-2488. doi:10.1001/jama.2013.6959.
Thalamic Stroke with Normal CTA
Artery of Percheron Infarction: Imaging Patterns and Clinical Spectrum
Dual Anti-Platelet
Assoc. with higher chance of hemorrhage (Cucchiara B, Kasner SE, Tanne D, et al. Factors associated with intracerebral hemorrhage after thrombolytic therapy for ischemic stroke: pooled analysis of placebo data from the Stroke-Acute Ischemic NXY Treatment (SAINT) I and SAINT II Trials. Stroke. 2009 Sep;40(9):3067-72.)
Isolated hand or finger palsy
is
not included in the NIH Stroke
Scale, which is a weakness of the
tool because this type of palsy is
a disabling clinical condition and
could be an indicator of acute
ischemic stroke. (West J Emerg
Med 2018;19[3]:524; http://bit.
ly/2OCzVW5.)
The incidence of acute stroke
with isolated hand paresis is not
known, but it appears to constitute
from 0.83 percent to 1.5 percent of
all ischemic strokes. (Cerebrovasc
Dis Extra 2013;3[1]:65; http://bit.ly/
2OzrQ4k.) A paralysis of the hand
and fingers without sensory deficit is
due to cortical infarction of the precentral
dial knob, a segment of the
precentral gyrus that projects to
the middle genu of the central sulcus.
(Neurology 2001;56[12]:1677.)
Embolic mechanisms are more often
associated with small cortical infarction
associated with isolated hand
or finger palsy. (West J Emerg Med
2018;19[3]:524; http://bit.ly/2OCzVW5;
Neurology 2001;56[12]:1677.)
Clinicians must consider that patients
with isolated hand palsy may
have an alternative explanation, including
a history of pain suggesting
vasculitis, waking from sleep with
the deficit (compression), a fall
(trauma), and shoulder (neuralgic
amyotrophy) or neck pain with radicular
symptoms. The patient
should be aggressively evaluated
and treated for acute ischemic
stroke if these findings are absent.
(West J Emerg Med 2018;19[