Note this drug is deader than a doornail. This page is maintained solely for archival purposes.
Protein C / Xigris /
Guidelines for the use of Xigris®
(Drotrecogin alfa (activated) rhu)
ELI LILLY AUSTRALIAN CLINICAL ADVISORY BOARD
In recognition of the unique properties of Xigris® (drotrecogin alfa (activated), also referred to as human recombinant activated protein C), Eli Lilly Australia established an Australian Clinical Advisory Board (see Appendix 2 for the composition of the Board). One of the goals of the board was to develop practical clinical guidelines for the appropriate use of Xigris. These guidelines are the result of the Boards deliberations. The Board encourages use of these guidelines to aid in patient selection for treatment with Xigris in your hospital.
The Board recommends use of Xigris in patients with severe sepsis at a higher risk of death, in accordance with the TGA approved indication. The guidelines characterize patients using a stepwise algorithm involving identification of (i) presence of infection, (ii) presence of systemic inflammatory response syndrome (SIRS) criteria and (iii) presence of at least one organ dysfunction using standardised criteria or a modified pre-infusion APACHE II score to select appropriate patients for Xigris therapy.
The board strongly recommends that patients considered for treatment with Xigris are admitted to an intensive care unit. All patients should receive full resuscitation as required with fluids, vasopressor agents, inotropes, ventilatory assistance, surgery, percutaneous drainage and antibiotics before consideration of Xigris therapy.
The appendices contain summary tables that assisted the Board in the development of these guidelines. A comprehensive list of contra-indications is listed.
After examination of the available evidence, the Board believes that appropriate use of Xigris improves survival in patients with severe sepsis at a high risk of death and represents a cost effective addition to treatment for patients with severe sepsis.
Patients should be admitted to the Intensive Care Unit and receive full resuscitation including any appropriate surgical intervention for source control. Four hours of resuscitation is considered a reasonable guide. If objective improvement in organ function occurs delay the administration of Xigris. Patients that show marked improvement have a lower risk of death.
Severe sepsis at a high risk of death is defined as:
1)The patient is infected:
1. Known infection.
2. Suspected infection
· White blood cells in usually sterile fluid.
· Perforated viscus.
· X-ray evidence of pneumonia and sputum production.
3. Syndrome associated with a high risk of infection.
And;
2) Presence of systemic inflammatory response manifested by > 3 out of the following criteria:
1. Core temperature ³ 38°C or £ 36°C.
2. Heart rate > 90 beats per minute in the absence of known illness, heart rhythm abnormality causing tachycardia or patients treated with a drug known to prevent tachycardia.
3. Respiratory rate ³ 20 breaths per minute or pCO2 £ 32mmHg or mechanical ventilation
4. White cell count > 12,000/mm3, or < 4000/mm3 or > 10% immature neutrophils.
And;
3)The patient has 2 or more organ system dysfunctions related to the infective process (organ dysfunction as defined below).
RESPIRATORY:
Respiratory failure should be due to lung injury/ARDS secondary to sepsis. The patient is expected to be on ventilatory support.
Ratio PaO2 to FiO2 £ 250 in the presence of other organ dysfunctions
Or
Ratio PaO2 to FiO2 £ 200.
CIRCULATORY:
Arterial systolic blood pressure £ 90mmHg or a mean arterial blood pressure
£ 70mmHg for 4 hours despite adequate fluid resuscitation or, the need for inotropic/vasopressor support to maintain arterial pressure above these levels.
RENAL:
Urine volume £ 0.5 ml/kg/hr for 4 hours in the absence of chronic renal failure, and in the presence of appropriate resuscitation. (The ANZICS CTG data indicates this definition of organ failure is associated with a 42% mortality rate independent of the creatinine level).
METABOLIC:
Arterial pH £ 7.30, or base deficit ³ 5.0 mmol/L, and a plasma lactate ³ 3mmol/L over 4 hours not explained by drug therapy, fitting or dialysis.
HAEMATOLOGICAL:
A platelet count of 80,000 mm3 or less, or a fall in platelet count of 50% over 3 days in the absence of factors apart from sepsis that can cause a falling or low platelet count.
Please refer to precautions regarding platelet count
Or:
The patient has a single organ system dysfunction (as above) and a modified pre-infusion APACHE II score >25.
The modified pre-infusion APACHE II score is the worst score in the 24-hours preceding the assessment, and may include data collected outside of the ICU if available (a scoring chart is attached to assist in calculating the score).
CONTRAINDICATIONS TO THE ADMINISTRATION OF XIGRIS
1. Pregnancy
2. Active internal bleeding
3. Recent (within 3 months) haemorrhagic stroke
4. Recent (within 2 months) intracranial or intraspinal surgery, severe head trauma requiring hospitalisation
5. Trauma patients with increased risk of life-threatening bleeding
6. Patients with an epidural catheter
7. Patients with intracranial neoplasm or mass lesion
PRECAUTIONS
Xigris increases the risk of bleeding. In the following conditions the risk of administration should be weighed against the anticipated benefits.
1. Platelet count < 30,000/mm3, even if the platelet count is increased after platelet transfusion
2. Concurrent heparin therapy greater than or equal to 15 international units per kilogram per hour
3. Recent (within 6 weeks) gastrointestinal bleeding
4. Recent administration (within 3 days) of thrombolytic therapy
5. Recent administration (within 7 days) of oral anticoagulants or glycoprotein IIb/IIIa inhibitors
6. Recent administration (within 7 days) of aspirin greater than 650 mg per day or other platelet inhibitors
7. Recent (within 3 months) ischaemic stroke
8. Patients with intracranial arteriovenous malformations or aneurysms
9. Know bleeding diathesis except for the coagulopathy associated with sepsis
10. Chronic severe hepatic disease
11. Any condition where the physician think the risk of bleeding is high
12. Conditions that increased the risk of bleeding: surgery requiring general or spinal anaesthesia within 12 hours before the infusion.
13. Human immunodeficiency virus infection in association with a last known CD4 count of < 50/mm3.
14. Chronic renal failure requiring haemodialysis or peritoneal dialysis
15. Acute pancreatitis with no established source of infection.
Xigris use in the PROWESS study was limited to patients 18 years of age and over. Use in children requires special consideration of the risks and benefits associated with Xigris There is insufficient data to support a dosage recommendation in patients with end stage renal disease.
The Board does not recommend use in the terminally ill patient or in patients in whom death appears imminent. There are no studies on the use of Xigris in lactating patients, or on the transfer into breast milk.
SPECIAL CONSIDERATIONS
Procedures during Xigris infusion
Xigris infusion should be stopped 2 hours before percutaneous procedures where there is a risk of bleeding. Xigris may be restarted immediately after uncomplicated less invasive procedures
Xigris infusion should be stopped 2 hours before major invasive procedures or surgery. Xigris infusion may be started 12 hours after a major invasive procedures or surgery if adequate haemostasis has been achieved.
Purpura fulminans, or meningococcal septicaemia
This form of severe sepsis carries a very high mortality and morbidity rate. Xigris should be considered for patients with purpura fulminans syndrome, however bleeding risk associated with thrombocytopaenia, coagulation abnormalities, high rate of cerebral haemorrhage and gastrointestinal haemorrhage must be assessed. Xigris should only be used where the clinician considers the benefits outweigh the risk.
Here’s a shortened version of our criteria.
Anant Murthy MD FRCPC
Intensivist, Trillium Health Centre
Mississauga ON Canada
Activated protein C (drotrecogin alfa activated) decreased mortality to 24.7% from 30.8% (NEJM 344:699, 2001. NNT 16). Bleeding was increased (3.5% vs 2%) and risk of intracranial hemorrhage remains unclear. In this study, basic criteria were: 1. infection, 2. 3+ SIRS signs within 24h, 3.
1+ systems dysfunction for <24h, 4. treatment <24h after criteria met.
Suggested criteria (Can J Infect Dis 13:365, 2002) Patients should have 3 or 4 signs of SIRS 1. core temperature <=36 or >=38
2. HR>90
3. RR>=20 or PaCO2<=32 or ventilation 4. WBC >=12 or <=4 or >10% immature neutrophils
Patients should have evidence of serious infection (appropriately treated with antibiotics).
1. positive cultures
2. WBC in a normally sterile fluid 3. perforated viscus
4. radiographic evidence of pneumonia with purulent sputum 5. purpura fulminans
6. syndrome (e.g. ascending cholangitis, necrotizing fasciitis) associated with a high risk of infection
Patients should have 2 or more acute organ dysfunctions (not explained by disease process or therapy) that developed within approximately the last 48h.
vasopressors or SBP<90/MAP<70 for 1h despite fluids urine<0.5mL/kg/h for 1h despite fluids P/F<250
Platelets decreased by 50% or <80 in the last 3 days Lactate > 1.5 x upper limit of normal AND pH<7.30 or base deficit
APC absolute contraindications include endstage disease with expected survival less than 28 days, active internal/postoperative bleeding, intracranial mass lesion or herniation, recent (<24h) epidural/spinal catheterization, recent severe head trauma requiring hospitalization, trauma with an increased risk of life threatening bleeding, recent (<2 months) intracranial/intraspinal surgery, and recent (<3 months) hemorrhagic stroke.
APC relative contraindications include anticoagulants (INR=> 3, ATIII>10000u within 12h, heparin>15U/kg/h or therapeutic LMWH within 8h,), fibrinolytics (within 24h), antiplatelet agents (ASA >650mg/d within 3d) within 7d, coagulopathy (including liver disease and recent hemorrhage but excluding that related to sepsis), GI bleed (within 6 weeks unless cured), ischemic stroke (within 3 months), intracranial AVM/aneurysm, pregnancy, and lactation.
Age<18, transplant, hypercoagulable condition, AIDS with last known CD4<50, chronic renal failure, and weight>135kg should all cause caution.
Platelets less than 30 are a significant risk factor for intracranial hemorrhage due to APC, but also for death due to sepsis. The role of transfusion is uncertain.
APC should be held:
1h before central or arterial lines, or ETT or tracheostomy changes 2h before and 1h after thoracentesis or chest tube, lumbar or sinus puncture, or tracheostomy
2h before epidural insertion (and no resumption) 2h before and 12h after epidural removal 2h before and 12h after major surgery
The dose of APC is 24µg/kg/hx96h. PT, PTT, and CBC should be checked daily.
Protein C
PROWESS Trial
Bernard GR, Vincent JLV, LaTerre PF, et al. Efficacy and safety of recombinant human activated protein C for severe sepsis. N Engl J Med. 2001;344:699-709
Very different data submitted to FDA. Only severely ill patients benefited, study drug changed halfway through as were the entry criteria.
Contraindications to protein c
active internal bleeding recent hemorrhagic stroke (3 months) recent (2 months) head trauma or head/spine surg trauma at risk for bleeding epidural catheter intrcranial mass or neoplasm plt <30000 9transfusion of plt will not help this) indications are sepsis c apache>25 or multi-organ failure 12 hours after surgical procedure normal hemostasis resums 2 hours after cessation of infusion get table iii
subgroup analysis shows large part of the benefit is in patients with severe CAP especially strep pneumo (Crit Care Med 2005;33(5):952)
ADDRESS Trial
Drotrecogin Alfa (Activated) for Adults with Severe Sepsis and a Low Risk of Death: no benefit with large bleeding risk (NEJM 2005;353(13):1332-1341)
EXPRESS
no diff if heparin is added
One stage coagulation assays such as factor VIII, IX and XI assays are influenced by drotrecogin action, as also is the aPTT assay. There are other markers for septic coagulopathy and also for drotrecogin-induced bleeding that can be used to monitor therapy,and they should be used together, sequentially. The simplest of these are the INR and platelet count. A platelet count under 30 000/ mm3, even with improvement after transfusion, is a clear indication to stop the infusion or to accept the increased risk of severe bleeding, but I believe that higher values, with a fast descending curve can also be a good reason for thinking seriously about the treatment, especially if associated coagulation tests are also altered. Sequential platelet counts are an useful follow up tool for several kinds of coagulopathy.INR is largely dependant on the extrinsic pathway factors which are not influenced by drotrecogin, and it can also be a reasonably good way to detect a severe derangement that may indicate it´s time to stop the drotrecogin infusion, especially if taken together with platelet counts and other coag tests.
–Claudia Teles
Expert Panel on Xigris
Editorial
Increasing evidence that the risks of rhAPC may outweigh its benefits
Peter Q. Eichacker1 and Charles Natanson1
(1) Department of Critical Care Medicine, Building 10, Room 7D43, Clinical Center, National Institutes of Health, MD 20892 Bethesda, USA
Peter Q. EichackerEmail: peichacker@mail.cc.nih.gov
Received: 24 January 2007 Accepted: 24 January 2007 Published online: 15 February 2007
Without Abstract This editorial refers to the articles available at: http://eresources.library.mssm.edu:2079/10.1007/s00134-007-0554-x and http://eresources.library.mssm.edu:2079/10.1007/s00134-007-0555-9
Sir: The surveys by Kanji et al. and Bertolini et al. in this issue of Intensive Care Medicine are important in providing further insights into the increased bleeding risks associated with recombinant human activated protein C (rhAPC) in clinical practice [1, 2]. Following approval of rhAPC in 2001, early surveys and post-marketing studies suggested that the risk of bleeding during clinical use might be greater than was noted in the original phase-3 trial [Recombinant Human Activated protein C Worldwide Evaluation in Severe Sepsis (PROWESS) trial; 3, 4, 5, 6]. Even more concerning, two additional randomized controlled sepsis trials conducted post-approval have failed to show any significant benefit with rhAPC [7, 8]. In light of increasing experience with rhAPC, the critical care community should reconsider whether the bleeding risks associated with this agent outweigh its purported benefit.
Increased bleeding with rhAPC has been a consistent finding in all sepsis trials conducted to date. In both PROWESS (n = 1,690) and a second randomized controlled trial in adults [Administration of Drotrecogin Alfa (Activated) During Early Severe Sepsis (ADDRESS; n = 2,640)], serious bleeding was greater during rhAPC infusion than placebo (2.4 vs. 1.0 and 2.4 vs. 1.2%, respectively, p = 0.02 for both comparisons) [6, 7, 8, 9]. Furthermore, in surgical patients with one organ injury a subgroup particularly susceptible to the risk of bleeding mortality rates were increased with treatment in both trials prompting a black box restriction on the rhAPC label [10]. In an open-label trial of rhAPC [Extended Evaluation of rhAPC trial (ENHANCE; n = 2,378)], despite employing inclusion and exclusion criteria similar to PROWESS, serious bleeding during drug administration was greater (3.6%) than in the two controlled trials [11]. Finally, in a controlled trial in children [Resolution of Organ Failure in Pediatric Patients with Severe Sepsis (RESOLVE; n = 399)], although the incidence of overall serious hemorrhage did not differ in study groups, a troubling increase in the number of intracerebral hemorrhages was associated with infusion of rhAPC (4 with treatment vs. 1 with placebo) [8]. Due in part to this finding and in part to lack of efficacy, the trial was stopped early.
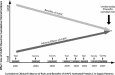
The risk of hemorrhage with rhAPC during clinical use has been consistently greater than reported in controlled trials of the agent (Fig. 1). In two early surveys in septic adults, one a retrospective observational study performed in 2002 (MERCURY; n = 274) and the other encompassing medical utilization evaluations done in 2002 and 2003 (n = 599) by the Voluntary Health Association and University Health System Consortium (VHA and UHC), the incidences of serious bleeding with rhAPC treatment were 4.0 and 4.7%, respectively [3, 4, 5]. In the more recent survey by G. Bertolini the incidence of serious bleeding was 4.6% [2]. S. Kanji’s survey [1] reports the risk of bleeding during rhAPC use as 7.3%, three times greater than in the original PROWESS trial [6]. In addition to increasing the risk of bleeding, rhAPC infusion in these clinical use studies was associated with a relatively high fatal event rate and an increased need to discontinue rhAPC due to bleeding (Fig. 1). Fig. 1 Incidence (percentage of patients) during rhAPC infusion of serious hemorrhage (top panel), a drug related fatal event (middle panel), or termination of treatment due to a drug-related adverse event (lower panel). Data are from three randomized controlled trials and one open-label trial and four clinical use surveys [1, 2, 3, 4, 5, 6, 7, 8, 11]. The full titles are provided in the text of the editorial for each trial or survey While surveys [1, 2, 3, 4, 5] indicate that the bleeding risk with rhAPC during clinical use is greater than originally estimated in PROWESS [6], the efficacy of rhAPC noted in this early trial has not been reproduced in subsequent trials [7, 8]. Neither of the controlled trials that followed PROWESS (i.e., ADDRESS and RESOLVE) noted any significant benefit with rhAPC, regardless of underlying severity of disease (Fig. 2) [6, 7, 8]. Across all trials that compared rhAPC to placebo (enrolling approximately 5,000 patients), only two subgroups from one study demonstrated benefit (i.e., the two highest APACHE-II quartiles from the PROWESS trial) [12]. These two subgroups included only 414 patients receiving rhAPC (i.e., 17% of all patients receiving the agent in controlled trials). Notably, the later ADDRESS trial (n = 2,639 patients) also enrolled high-risk patients [i.e., APACHE-II score ≥ 25 (n = 324) receiving either rhAPC or placebo] but failed to reproduce the finding of efficacy. In fact, for high-risk patients the treatment effect in ADDRESS was on the side of harm, opposite to and significantly different from the effect of rhAPC in PROWESS (Fig. 2) [12]. In PROWESS the decision to treat patients with rhAPC was not based on the APACHE score. Since these scores were determined retrospectively using the most extreme values obtained over a 24-h period before the drug was administered, it is unknown if this even represents a clinically definable population. Conversely, in ADDRESS, APACHE-II scores (or other measure of risk) were calculated prospectively and used to determine whom to treat. This is similar to what would occur in clinical practice, perhaps making the results more relevant. Casting further doubt on efficacy in clinical practice, mortality rates from surveys describing patients treated with rhAPC [1, 4, 5] have been consistently higher than was seen in PROWESS [6], even after stratifying for severity of illness (Fig. 3). While this does not constitute proof of harm, it is worrisome. Additionally, while rhAPC was approved for high-risk patients based on PROWESS, in the survey by Kanji et al. patients with four to five injured organs and a high risk of death had a marked increase in the risk of serious bleeding with rhAPC [1]. At this time therefore, it appears that it would be difficult for clinicians to confidently identify a population of patients that is likely to benefit from rhAPC. Fig. 2 Effect of rhAPC on the relative risk of death in three controlled trials. Adult patients in the PROWESS [6] and ADDRESS [7] were stratified based on whether their admission APACHE-II scores were greater or less than 25 Fig. 3 The mortality rate in patients treated with rhAPC, stratified by number of organs injured in the PROWESS trial [6], VHA/UHC survey [4, 5], and Canadian [1] survey. Despite similar numbers of injured organs, mortality rates were greater in patients treated with rhAPC in both clinical surveys compared with the PROWESS trial. Similar data was not available for analysis from the other controlled trials and clinical surveys While the apparent efficacy of rhAPC in PROWESS [6] has not been reproduced in subsequent trials [7, 8], an increased risk of bleeding has been confirmed in controlled trials and documented at even higher rates in clinical use (Figs. 1, 2) [1, 2, 3, 4, 5]. Why this risk appears more pronounced than during the original PROWESS trial is likely multi-factorial [13, 14]. For example, investigating rhAPC in larger of numbers of patients in the ADDRESS trial was necessary to clearly document the agent’s risk in surgical patients. Also, during controlled trials that are designed primarily to show the efficacy of an agent, exclusion criteria are frequently applied that minimize the occurrence of adverse effects. During clinical use of an agent, such exclusion criteria may not be applied as rigorously and the incidence of adverse events would be expected to increase. Overall, however, our growing experience with rhAPC represents a change in our understanding of the risk-to-benefit ratio of rhAPC (Fig. 4). Further randomized controlled trials are necessary to demonstrate whether patients can be prospectively identified that benefit from rhAPC and if such benefit is large enough to warrant the use of rhAPC in clinical practice where the risk of bleeding appears substantial. Fig. 4 Our understanding of the relative size of the beneficial and adverse effects of rhAPC. This is based on accumulating evidence from controlled trials and clinical surveys starting in 2001 following completion of the PROWESS trial and licensing of rhAPC for clinical use. Since the original beneficial effects of rhAPC have not been confirmed in subsequent trials, but the incidence of bleeding has persisted in trials and increased in clinical use, it is unclear presently whether the benefit of this agent outweighs its risks
References
1. Kanji S, Perreault MM, Chant C, Williamson D, Burry L (2007) Evaluating the use of Drotrecogin alfa (activated) in adult severe sepsis: a Canadian multicenter observation study. Intensive Care Med DOI 10.1007/s00134-007-0555-9 2. Bertolini G, Rossi C, Anghileri A, Livigni S, Addis A, Poole D (2007) Use of drotrecogin alfa (activated) in Italian intensive care units. The results of a nationwide survey. Intensive Care Med DOI 10.1007/s00134-007-0554-x 3. Steingrub J, Sanchez P, Zeckel M, Bates B, Qualy R (2003) Safety of drotrecogin alfa (activated): results of MERCURY, a retrospective multicenter observation study. Crit Care Med 31:A117 4. Tanzi M (2004) Use of drotrecogin alfa (activated) (Xigris) for the treatment of severe sepsis: medical use evaluation by Novation, the supply company of VHA and UHC. Novation 5. Deans KJ, Minneci PC, Banks SM, Natanson C, Eichacker PQ (2004) Substantiating the concerns about recombinant human activated protein C use in sepsis. Crit Care Med 32:25422543 6. Bernard GR, Vincent JL, Laterre PF, LaRosa SP, Dhainaut JF, Lopex-Rodriguez A, Steingrub JS, Garber GE, Helterbrand JD, Ely EW et al. (2001) Efficacy and safety of human activated protein C for severe sepsis. N Engl J Med 344:699709 7. Abraham E, Laterre F, Garg R et al. (2005) Drotrecogin alfa (activated) for adults with severe sepsis and a low risk of death. N Engl J Med 353:13321341 8. Eisenberg P. Discontinuation of study F1K-MC-EVBP, investigation of the efficacy and safety of Drotrecogin alfa (activated) in pediatric severe sepsis (letter). http://eresources.library.mssm.edu:2906/medwatch/SAFETY/2005/xigris_dearhcp_4-21-05.htm. Accessed 5 July 2005 9. Eichacker PQ, Natanson C (2003) Recombinant human activated protein C in sepsis: inconsistent trial results, an unclear mechanism of action, and safety concerns resulted in labeling restrictions and the need for phase IV trials. Crit Care Med 31:S94S96 10. Eisenberg P. Important Drug Warning (letter) http://eresources.library.mssm.edu:2906/medwatch/SAFETY/2005/xigris_DHCP.htm. Accessed 5 July 2005 11. Vincent JL, Bernard GR, Beale R, Doig C, Putenson C, Dhainaut JF, Artigas A, Fumagelli R, Macias W, Wright T, Wong K, Sundin DP, Turlo MA, Janes J, for the ENHANCE Study Group (2005) Drotrecogin alfa (activated) treatment in severe sepsis from the Global Open-label Trial ENHANCE: further evidence for survival and safety. Crit Care Med 33:22662277 12. Eichacker PQ, Danner RL, Suffredini AF, Cui X, Natanson C (2005) Reassessing recombinant human activated protein C for sepsis: time for a new randomized trial. Crit Care Med 33:24262428 13. Hunter D (2006) First gather the data. N Engl J Med 354:329331 14. Vlahakes GJ (2006) The value of phase 4 clinical testing. N Engl J Med 354:413415
Cochrane Database SR
2008 Jan 23(1)
no evidence suggesting APC should be used at all
Assoc with high risk of bleeding
| | |